 |
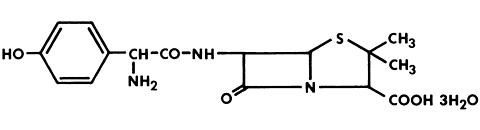
Augmentin is an oral antibacterial combination consisting of the semisynthetic antibiotic amoxicillin and the (beta)-lactamase inhibitor, clavulanate potassium (the potassium salt of clavulanic acid). Amoxicillin is an analog of ampicillin, derived from the basic penicillin nucleus, 6-aminopenicillanic acid. The amoxicillin molecular formula is C 16 H 19 N 3 O 5 S·3H 2 O and the molecular weight is 419.46. Chemically, amoxicillin is ( 2S,5R,6R )-6-[( R )-(-)-2-Amino-2-( p -hydroxyphenyl)acetamido] -3,3-dimethyl-7-oxo-4-thia-1-azabicyclo[3.2.0]heptane-2- carboxylic acid trihydrate and may be represented structurally as:
 |
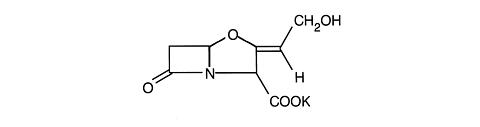
Clavulanic acid is produced by the fermentation of Streptomyces clavuligerus. It is a (beta)-lactam structurally related to the penicillins and possesses the ability to inactivate a wide variety of (beta)-lactamases by blocking the active sites of these enzymes. Clavulanic acid is particularly active against the clinically important plasmid mediated (beta)-lactamases frequently responsible for transferred drug resistance to penicillins and cephalosporins. The clavulanate potassium molecular formula is C 8 H 8 KNO 5 and the molecular weight is 237.25. Chemically clavulanate potassium is potassium (Z)-( 2R,5R )-3-(2-hydroxyethylidene) -7-oxo-4-oxa-1-azabicyclo [3.2.0]-heptane-2-carboxylate and may be represented structurally as:
 |
Inactive Ingredients: Powder for Oral Suspension--Colloidal silicon dioxide, flavorings (See HOW SUPPLIED ), succinic acid, xanthan gum, and one or more of the following: aspartame·, hydroxypropyl methylcellulose, mannitol, silica gel, silicon dioxide and sodium saccharin. Chewable Tablets--Colloidal silicon dioxide, flavorings (See HOW SUPPLIED ), magnesium stearate, mannitol and one or more of the following: aspartame·, D&C Yellow No. 10, FD&C Red No. 40, glycine, sodium saccharin and succinic acid.
·See PRECAUTIONS --Information for Patients.
Each 125 mg chewable tablet and each 5 mL of reconstituted Augmentin 125 mg/5 mL oral suspension contains 0.16 mEq potassium. Each 250 mg chewable tablet and each 5 mL of reconstituted Augmentin 250 mg/5 mL oral suspension contains 0.32 mEq potassium. Each 200 mg chewable tablet and each 5 mL of reconstituted Augmentin 200 mg/5 mL oral suspension contains 0.14 mEq potassium. Each 400 mg chewable tablet and each 5 mL of reconstituted Augmentin 400 mg/5 mL oral suspension contains 0.29 mEq of potassium.
Amoxicillin and clavulanate potassium are well absorbed from the gastrointestinal tract after oral administration of Augmentin. Dosing in the fasted or fed state has minimal effect on the pharmacokinetics of amoxicillin. While Augmentin can be given without regard to meals, absorption of clavulanate potassium when taken with food is greater relative to the fasted state. In one study, the relative bioavailability of clavulanate was reduced when Augmentin was dosed at 30 and 150 minutes after the start of a high fat breakfast. The safety and efficacy of Augmentin have been established in clinical trials where Augmentin was taken without regard to meals.
Oral administration of single doses of 400 mg Augmentin chewable tablets and 400 mg/5 mL suspension to 28 adult volunteers yielded comparable pharmacokinetic data:
|
||||||||||||||||||||||||||||||
Oral administration of 5 mL of Augmentin 250 mg/5 mL suspension or the equivalent dose of 10 mL Augmentin 125 mg/5 mL suspension provides average peak serum concentrations approximately 1 hour after dosing of 6.9 µg/mL for amoxicillin and 1.6 µg/mL for clavulanic acid. The areas under the serum concentration curves obtained during the first 4 hours after dosing were 12.6 µg.hr./mL for amoxicillin and 2.9 µg.hr./mL for clavulanic acid when 5 mL of Augmentin 250 mg/5 mL suspension or equivalent dose of 10 mL of Augmentin 125 mg/5 mL suspension was administered to adult volunteers. One Augmentin 250 mg chewable tablet or 2 Augmentin 125 mg chewable tablets are equivalent to 5 mL of Augmentin 250 mg/5 mL suspension and provide similar serum levels of amoxicillin and clavulanic acid.
Amoxicillin serum concentrations achieved with Augmentin are similar to those produced by the oral administration of equivalent doses of amoxicillin alone. The half-life of amoxicillin after the oral administration of Augmentin is 1.3 hours and that of clavulanic acid is 1.0 hour. Time above the minimum inhibitory concentration of 1.0 µg/mL for amoxicillin has been shown to be similar after corresponding q12h and q8h dosing regimens of Augmentin in adults and children.
Approximately 50% to 70% of the amoxicillin and approximately 25% to 40% of the clavulanic acid are excreted unchanged in urine during the first 6 hours after administration of 10 mL of Augmentin 250 mg/5 mL suspension.
Concurrent administration of probenecid delays amoxicillin excretion but does not delay renal excretion of clavulanic acid.
Neither component in Augmentin is highly protein-bound; clavulanic acid has been found to be approximately 25% bound to human serum and amoxicillin approximately 18% bound.
Amoxicillin diffuses readily into most body tissues and fluids with the exception of the brain and spinal fluid. The results of experiments involving the administration of clavulanic acid to animals suggest that this compound, like amoxicillin, is well distributed in body tissues.
Two hours after oral administration of a single 35 mg/kg dose of Augmentin suspension to fasting children, average concentrations of 3.0 µg/mL of amoxicillin and 0.5 µg/mL of clavulanic acid were detected in middle ear effusions.
Microbiology Amoxicillin is a semisynthetic antibiotic with a broad spectrum of bactericidal activity against many gram-positive and gram-negative microorganisms. Amoxicillin is, however, susceptible to degradation by (beta)-lactamases and, therefore, the spectrum of activity does not include organisms which produce these enzymes. Clavulanic acid is a (beta)-lactam, structurally related to the penicillins, which possesses the ability to inactivate a wide range of (beta)-lactamase enzymes commonly found in microorganisms resistant to penicillins and cephalosporins. In particular, it has good activity against the clinically important plasmid mediated (beta)-lactamases frequently responsible for transferred drug resistance.
The formulation of amoxicillin and clavulanic acid in Augmentin protects amoxicillin from degradation by (beta)-lactamase enzymes and effectively extends the antibiotic spectrum of amoxicillin to include many bacteria normally resistant to amoxicillin and other (beta)-lactam antibiotics. Thus, Augmentin possesses the distinctive properties of a broad-spectrum antibiotic and a (beta)-lactamase inhibitor.
Amoxicillin/clavulanic acid has been shown to be active against most strains of the following microorganisms, both in vitro and in clinical infections as described in the section.
Staphylococcus aureus ((beta)-lactamase and non-(beta)-lactamase producing)§
§Staphylococci which are resistant to methicillin/oxacillin must be considered resistant to amoxicillin/clavulanic acid.
Enterobacter species (Although most strains of Enterobacter species are resistant in vitro, clinical efficacy has been demonstrated with Augmentin in urinary tract infections caused by these organisms.)
Escherichia coli ((beta)-lactamase and non-(beta)-lactamase producing)
Haemophilus influenzae ((beta)-lactamase and non-(beta)-lactamase producing)
Klebsiella species (All known strains are (beta)-lactamase producing.)
Moraxella catarrhalis ((beta)-lactamase and non-(beta)-lactamase producing)
The following in vitro data are available, but their clinical significance is unknown .
Amoxicillin/clavulanic acid exhibits in vitro minimal inhibitory concentrations (MICs) of 0.5 µg/mL or less against most (>/=90%) strains of Streptococcus pneumoniae II ; MICs of 0.06 µg/mL or less against most (>/=90%) strains of Neisseria gonorrhoeae; MICs of 4 µg/mL or less against most (>/=90%) strains of staphylococci and anaerobic bacteria; and MICs of 8 µg/mL or less against most (>/=90%) strains of other listed organisms. However, with the exception of organisms shown to respond to amoxicillin alone, the safety and effectiveness of amoxicillin/clavulanic acid in treating clinical infections due to these microorganisms have not been established in adequate and well-controlled clinical trials.
II Because amoxicillin has greater in vitro activity against Streptococcus pneumoniae than does ampicillin or penicillin, the majority of S. pneumoniae strains with intermediate susceptibility to ampicillin or penicillin are fully susceptible to amoxicillin.
Enterococcus faecalis ¶
Staphylococcus epidermidis ((beta)-lactamase and non-(beta)-lactamase producing)
Staphylococcus saprophyticus ((beta)-lactamase and non-(beta)-lactamase producing)
Streptococcus pneumoniae ¶**
Streptococcus pyogenes ¶**
viridans group Streptococcus ¶**
Eikenella corrodens ((beta)-lactamase and non-(beta)-lactamase producing)
Neisseria gonorrhoeae ¶ ((beta)-lactamase and non-(beta)-lactamase producing)
Proteus mirabilis ¶ ((beta)-lactamase and non-(beta)-lactamase producing)
Bacteroides species, including Bacteroides fragilis ((beta)-lactamase and non-(beta)-lactamase producing)
Fusobacterium species ((beta)-lactamase and non-(beta)-lactamase producing)
Peptostreptococcus species
¶Adequate and well-controlled clinical trials have established the effectiveness of amoxicillin alone in treating certain clinical infections due to these organisms.
** These are non-(beta)-lactamase-producing organisms and, therefore, are susceptible to amoxicillin alone.
Dilution Techniques : Quantitative methods are used to determine antimicrobial minimal inhibitory concentrations (MICs). These MICs provide estimates of the susceptibility of bacteria to antimicrobial compounds. The MICs should be determined using a standardized procedure. Standardized procedures are based on a dilution method 1 (broth or agar) or equivalent with standardized inoculum concentrations and standardized concentrations of amoxicillin/clavulanate potassium powder.
The recommended dilution pattern utilizes a constant amoxicillin/clavulanate potassium ratio of 2 to 1 in all tubes with varying amounts of amoxicillin. MICs are expressed in terms of the amoxicillin concentration in the presence of clavulanic acid at a constant 2 parts amoxicillin to 1 part clavulanic acid. The MIC values should be interpreted according to the following criteria:
RECOMMENDED RANGES FOR AMOXICILLIN/CLAVULANIC ACID SUSCEPTIBILITY TESTING
For gram-negative enteric aerobes:
|
For Staphylococcus ** and Haemophilus species
|
||||||||
For Streptococcus pneumoniae: Isolates should be tested using amoxicillin/clavulanic acid and the following criteria should be used:
|
A report of "Susceptible" indicates that the pathogen is likely to be inhibited if the antimicrobial compound in the blood reaches the concentration usually achievable. A report of "Intermediate" indicates that the result should be considered equivocal, and, if the microorganism is not fully susceptible to alternative, clinically feasible drugs, the test should be repeated. This category implies possible clinical applicability in body sites where the drug is physiologically concentrated or in situations where high dosage of drug can be used. This category also provides a buffer zone that prevents small uncontrolled technical factors from causing major discrepancies in interpretation. A report of "Resistant" indicates that the pathogen is not likely to be inhibited if the antimicrobial compound in the blood reaches the concentrations usually achievable; other therapy should be selected.
Standardized susceptibility test procedures require the use of laboratory control microorganisms to control the technical aspects of the laboratory procedures. Standard amoxicillin/clavulanate potassium powder should provide the following MIC values:
|
||||||||||||||||
Diffusion Techniques : Quantitative methods that require measurement of zone diameters also provide reproducible estimates of the susceptibility of bacteria to antimicrobial compounds. One such standardized procedure 2 requires the use of standardized inoculum concentrations. This procedure uses paper disks impregnated with 30 µg of amoxicillin/clavulanate potassium (20 µg amoxicillin plus 10 µg clavulanate potassium) to test the susceptibility of microorganisms to amoxicillin/clavulanic acid.
Reports from the laboratory providing results of the standard single-disk susceptibility test with a 30 µg amoxicillin/clavulanate potassium (20 µg amoxicillin plus 10 µg clavulanate potassium) disk should be interpreted according to the following criteria:
RECOMMENDED RANGES FOR AMOXICILLIN/CLAVULANIC ACID SUSCEPTIBILITY TESTING
For Staphylococcus §§ species and H. influenzae a :
|
For other organisms except S. pneumoniae b and N. gonorrhoeae c :
|
§§Staphylococci which are resistant to methicillin/oxacillin must be considered as resistant to amoxicillin/clavulanic acid.
a A broth microdilution method should be used for testing H. influenzae. Beta-lactamase negative, ampicillin-resistant strains must be considered resistant to amoxicillin/clavulanic acid.
b Susceptibility of S. pneumoniae should be determined using a 1 µg oxacillin disk. Isolates with oxacillin zone sizes of >/=20 mm are susceptible to amoxicillin/clavulanic acid. An amoxicillin/clavulanic acid MIC should be determined on isolates of S. pneumoniae with oxacillin zone sizes of </=19 mm.
c A broth microdilution method should be used for testing N. gonorrhoeae and interpreted according to penicillin breakpoints.
Interpretation should be as stated above for results using dilution techniques. Interpretation involves correlation of the diameter obtained in the disk test with the MIC for amoxicillin/clavulanic acid.
As with standardized dilution techniques, diffusion methods require the use of laboratory control microorganisms that are used to control the technical aspects of the laboratory procedures. For the diffusion technique, the 30 µg amoxicillin/clavulanate potassium (20 µg amoxicillin plus 10 µg clavulanate potassium) disk should provide the following zone diameters in these laboratory quality control strains:
|
Augmentin is indicated in the treatment of infections caused by susceptible strains of the designated organisms in the conditions listed below:
Lower Respiratory Tract Infections --caused by (beta)-lactamase-producing strains of Haemophilus influenzae and Moraxella (Branhamella) catarrhalis.
Otitis Media --caused by (beta)-lactamase-producing strains of Haemophilus influenzae and Moraxella (Branhamella) catarrhalis.
Sinusitis --caused by (beta)-lactamase-producing strains of Haemophilus influenzae and Moraxella (Branhamella) catarrhalis.
Skin and Skin Structure Infections --caused by (beta)-lactamase-producing strains of Staphylococcus aureus, Escherichia coli and Klebsiella spp.
Urinary Tract Infections --caused by (beta)-lactamase-producing strains of Escherichia coli, Klebsiella spp. and Enterobacter spp.
While Augmentin is indicated only for the conditions listed above, infections caused by ampicillin-susceptible organisms are also amenable to Augmentin treatment due to its amoxicillin content. Therefore, mixed infections caused by ampicillin-susceptible organisms and (beta)-lactamase-producing organisms susceptible to Augmentin should not require the addition of another antibiotic. Because amoxicillin has greater in vitro activity against Streptococcus pneumoniae than does ampicillin or penicillin, the majority of S. pneumoniae strains with intermediate susceptibility to ampicillin or penicillin are fully susceptible to amoxicillin and Augmentin. (See Microbiology subsection.)
Bacteriological studies, to determine the causative organisms and their susceptibility to Augmentin, should be performed together with any indicated surgical procedures.
Therapy may be instituted prior to obtaining the results from bacteriological and susceptibility studies to determine the causative organisms and their susceptibility to Augmentin when there is reason to believe the infection may involve any of the (beta)-lactamase-producing organisms listed above. Once the results are known, therapy should be adjusted, if appropriate.
Augmentin is contraindicated in patients with a history of allergic reactions to any penicillin. It is also contraindicated in patients with a previous history of Augmentin -associated cholestatic jaundice/hepatic dysfunction.
SERIOUS AND OCCASIONALLY FATAL HYPERSENSITIVITY (ANAPHYLACTIC) REACTIONS HAVE BEEN REPORTED IN PATIENTS ON PENICILLIN THERAPY. THESE REACTIONS ARE MORE LIKELY TO OCCUR IN INDIVIDUALS WITH A HISTORY OF PENICILLIN HYPERSENSITIVITY AND/OR A HISTORY OF SENSITIVITY TO MULTIPLE ALLERGENS. THERE HAVE BEEN REPORTS OF INDIVIDUALS WITH A HISTORY OF PENICILLIN HYPERSENSITIVITY WHO HAVE EXPERIENCED SEVERE REACTIONS WHEN TREATED WITH CEPHALOSPORINS. BEFORE INITIATING THERAPY WITH AUGMENTIN, CAREFUL INQUIRY SHOULD BE MADE CONCERNING PREVIOUS HYPERSENSITIVITY REACTIONS TO PENICILLINS, CEPHALOSPORINS OR OTHER ALLERGENS. IF AN ALLERGIC REACTION OCCURS, AUGMENTIN SHOULD BE DISCONTINUED AND THE APPROPRIATE THERAPY INSTITUTED. SERIOUS ANAPHYLACTIC REACTIONS REQUIRE IMMEDIATE EMERGENCY TREATMENT WITH EPINEPHRINE. OXYGEN, INTRAVENOUS STEROIDS AND AIRWAY MANAGEMENT, INCLUDING INTUBATION, SHOULD ALSO BE ADMINISTERED AS INDICATED.
Pseudomembranous colitis has been reported with nearly all antibacterial agents, including Augmentin, and has ranged in severity from mild to life-threatening. Therefore, it is important to consider this diagnosis in patients who present with diarrhea subsequent to the administration of antibacterial agents.
Treatment with antibacterial agents alters the normal flora of the colon and may permit overgrowth of clostridia. Studies indicate that a toxin produced by Clostridium difficile is one primary cause of "antibiotic associated colitis."
After the diagnosis of pseudomembranous colitis has been established, appropriate therapeutic measures should be initiated. Mild cases of pseudomembranous colitis usually respond to drug discontinuation alone. In moderate to severe cases, consideration should be given to management with fluids and electrolytes, protein supplementation and treatment with an antibacterial drug clinically effective against Clostridium difficile colitis
Augmentin should be used with caution in patients with evidence of hepatic dysfunction. Hepatic toxicity associated with the use of Augmentin is usually reversible. On rare occasions, deaths have been reported (less than 1 death reported per estimated 4 million prescriptions worldwide). These have generally been cases associated with serious underlying diseases or concomitant medications. (See CONTRAINDICATIONS and ADVERSE REACTIONS -- Liver )
General: While Augmentin possesses the characteristic low toxicity of the penicillin group of antibiotics, periodic assessment of organ system functions, including renal, hepatic and hematopoietic function, is advisable during prolonged therapy. A high percentage of patients with mononucleosis who receive ampicillin develop an erythematous skin rash. Thus, ampicillin class antibiotics should not be administered to patients with mononucleosis.
The possibility of superinfections with mycotic or bacterial pathogens should be kept in mind during therapy. If superinfections occur (usually involving Pseudomonas or Candida ), the drug should be discontinued and/or appropriate therapy instituted.
Information for the Patient: Augmentin may be taken every 8 hours or every 12 hours, depending on the strength of the product prescribed. Each dose should be taken with a meal or snack to reduce the possibility of gastrointestinal upset. Many antibiotics can cause diarrhea. If diarrhea is severe or lasts more than 2 or 3 days, call your doctor.
Make sure your child completes the entire prescribed course of treatment, even if he/she begins to feel better after a few days. Keep suspension refrigerated. Shake well before using. When dosing a child with Augmentin suspension (liquid), use a dosing spoon or medicine dropper. Be sure to rinse the spoon or dropper after each use. Bottles of Augmentin suspension may contain more liquid than required. Follow your doctor' instructions about the amount to use and the days of treatment your child requires. Discard any unused medicine.
Phenylketonurics: Each 200 mg Augmentin chewable tablet contains 2.1 mg phenylalanine; each 400 mg chewable tablet contains 4.2 mg phenylalanine; each 5 mL of either the 200 mg/5 mL or 400 mg/5 mL oral suspension contains 7 mg phenylalanine. The other Augmentin products do not contain phenylalanine and can be used by phenylketonurics. Contact your physician or pharmacist.
Drug Interactions: Probenecid decreases the renal tubular secretion of amoxicillin. Concurrent use with Augmentin may result in increased and prolonged blood levels of amoxicillin. Co-administration of probenecid cannot be recommended.
The concurrent administration of allopurinol and ampicillin increases substantially the incidence of rashes in patients receiving both drugs as compared to patients receiving ampicillin alone. It is not known whether this potentiation of ampicillin rashes is due to allopurinol or the hyperuricemia present in these patients. There are no data with Augmentin and allopurinol administered concurrently.
In common with other broad-spectrum antibiotics, Augmentin may reduce the efficacy of oral contraceptives.
Drug/Laboratory Test Interactions: Oral administration of Augmentin will result in high urine concentrations of amoxicillin. High urine concentrations of ampicillin may result in false-positive reactions when testing for the presence of glucose in urine using Clinitest®, Benedict' Solution or Fehling' Solution. Since this effect may also occur with amoxicillin and therefore Augmentin, it is recommended that glucose tests based on enzymatic glucose oxidase reactions (such as Clinistix® or Tes-Tape®) be used.
Following administration of ampicillin to pregnant women a transient decrease in plasma concentration of total conjugated estriol, estriol-glucuronide, conjugated estrone and estradiol has been noted. This effect may also occur with amoxicillin and therefore Augmentin.
Carcinogenesis, Mutagenesis, Impairment of Fertility: Long-term studies in animals have not been performed to evaluate carcinogenic potential.
Mutagenesis: The mutagenic potential of Augmentin was investigated in vitro with an Ames test, a human lymphocyte cytogenetic assay, a yeast test and a mouse lymphoma forward mutation assay, and in vivo with mouse micronucleus tests and a dominant lethal test. All were negative apart from the in vitro mouse lymphoma assay where weak activity was found at very high, cytotoxic concentrations.
Impairment of Fertility: Augmentin at oral doses of up to 1200 mg/kg/day (5.7 times the maximum human dose, 1480 mg/m 2 /day, based on body surface area) was found to have no effect on fertility and reproductive performance in rats, dosed with a 2:1 ratio formulation of amoxicillin:
Teratogenic effects. Pregnancy (Category B): Reproduction studies performed in pregnant rats and mice given Augmentin at oral dosages up to 1200 mg/kg/day, equivalent to 7200 and 4080 mg/m 2 /day, respectively (4.9 and 2.8 times the maximum human oral dose based on body surface area), revealed no evidence of harm to the fetus due to Augmentin. There are, however, no adequate and well-controlled studies in pregnant women. Because animal reproduction studies are not always predictive of human response, this drug should be used during pregnancy only if clearly needed.
Labor and Delivery: Oral ampicillin class antibiotics are generally poorly absorbed during labor. Studies in guinea pigs have shown that intravenous administration of ampicillin decreased the uterine tone, frequency of contractions, height of contractions and duration of contractions. However, it is not known whether the use of Augmentin in humans during labor or delivery has immediate or delayed adverse effects on the fetus, prolongs the duration of labor, or increases the likelihood that forceps delivery or other obstetrical intervention or resuscitation of the newborn will be necessary.
Nursing Mothers: Ampicillin class antibiotics are excreted in the milk; therefore, caution should be exercised when Augmentin is administered to a nursing woman.
Pediatric Use: Because of incompletely developed renal function in neonates and young infants, the elimination of amoxicillin may be delayed. Dosing of Augmentin should be modified in pediatric patients younger than 12 weeks (3 months). (See DOSAGE AND ADMINISTRATION-Pediatric.)
Augmentin is generally well tolerated. The majority of side effects observed in clinical trials were of a mild and transient nature and less than 3% of patients discontinued therapy because of drug-related side effects. From the original premarketing studies, where both pediatric and adult patients were enrolled, the most frequently reported adverse effects were diarrhea/loose stools (9%), nausea (3%), skin rashes and urticaria (3%), vomiting (1%) and vaginitis (1%). The overall incidence of side effects, and in particular diarrhea, increased with the higher recommended dose. Other less frequently reported reactions include: abdominal discomfort, flatulence and headache.
In pediatric patients (aged 2 months to 12 years), one U.S./Canadian clinical trial was conducted which compared Augmentin 45/6.4 mg/kg/day (divided q12h) for 10 days versus Augmentin 40/10 mg/kg/day (divided q8h) for 10 days in the treatment of acute otitis media. A total of 575 patients were enrolled, and only the suspension formulations were used in this trial. Overall, the adverse event profile seen was comparable to that noted above. However, there were differences in the rates of diarrhea, skin rashes/urticaria, and diaper area rashes. (See CLINICAL STUDIES .)
The following adverse reactions have been reported for ampicillin class antibiotics:
Gastrointestinal Diarrhea, nausea, vomiting, indigestion, gastritis, stomatitis, glossitis, black "hairy" tongue, mucocutaneous candidiasis, enterocolitis, and hemorrhagic/pseudomembranous colitis. Onset of pseudomembranous colitis symptoms may occur during or after antibiotic treatment. (See .)
Hypersensitivity Reactions: Skin rashes, pruritus, urticaria, angioedema, serum sickness-like reactions (urticaria or skin rash accompanied by arthritis, arthralgia, myalgia and frequently fever), erythema multiforme (rarely Stevens-Johnson Syndrome) and an occasional case of exfoliative dermatitis (including toxic epidermal necrolysis) have been reported. These reactions may be controlled with antihistamines and, if necessary, systemic corticosteroids. Whenever such reactions occur, the drug should be discontinued, unless the opinion of the physician dictates otherwise. Serious and occasional fatal hypersensitivity (anaphylactic) reactions can occur with oral penicillin. (See .)
Liver A moderate rise in AST (SGOT) and/or ALT (SGPT) has been noted in patients treated with ampicillin class antibiotics but the significance of these findings is unknown. Hepatic dysfunction, including increases in serum transaminases (AST and/or ALT), serum bilirubin and/or alkaline phosphatase, has been infrequently reported with Augmentin . It has been reported more commonly in the elderly, in males, or in patients on prolonged treatment. The histologic findings on liver biopsy have consisted of predominantly cholestatic, hepatocellular, or mixed cholestatic-hepatocellular changes. The onset of signs/symptoms of hepatic dysfunction may occur during or several weeks after therapy has been discontinued. The hepatic dysfunction, which may be severe, is usually reversible. On rare occasions, deaths have been reported (less than 1 death reported per estimated 4 million prescriptions worldwide). These have generally been cases associated with serious underlying diseases or concomitant medications.
Renal Interstitial nephritis and hematuria have been reported rarely.
Hemic and Lymphatic Systems: Anemia, including hemolytic anemia, thrombocytopenia, thrombocytopenic purpura, eosinophilia, leukopenia and agranulocytosis have been reported during therapy with penicillins. These reactions are usually reversible on discontinuation of therapy and are believed to be hypersensitivity phenomena. A slight thrombocytosis was noted in less than 1% of the patients treated with Augmentin. There have been reports of increased prothrombin time in patients receiving Augmentin and anticoagulant therapy concomitantly.
Central Nervous System: Agitation, anxiety, behavioral changes, confusion, convulsions, dizziness, insomnia, and reversible hyperactivity have been reported rarely.
Most patients have been asymptomatic following overdosage or have experienced primarily gastrointestinal symptoms including stomach and abdominal pain, vomiting, and diarrhea. Rash, hyperactivity, or drowsiness have also been observed in a small number of patients.
In the case of overdosage, discontinue Augmentin , treat symptomatically, and institute supportive measures as required. If the overdosage is very recent and there is no contraindication, an attempt at emesis or other means of removal of drug from the stomach may be performed. A prospective study of 51 pediatric patients at a poison center suggested that overdosages of less than 250 mg/kg of amoxicillin are not associated with significant clinical symptoms and do not require gastric emptying. 3
Interstitial nephritis resulting in oliguric renal failure has been reported in a small number of patients after overdosage with amoxicillin. Renal impairment appears to be reversible with cessation of drug administration. High blood levels may occur more readily in patients with impaired renal function because of decreased renal clearance of both amoxicillin and clavulanate. Both amoxicillin and clavulanate are removed from the circulation by hemodialysis.
Pediatric Patients: Based on the amoxicillin component, Augmentin should be dosed as follows:
Neonates and infants aged < 12 weeks (3 months)
Due to incompletely developed renal function affecting elimination of amoxicillin in this age group, the recommended dose of Augmentin is 30 mg/kg/day divided q12h, based on the amoxicillin component. Clavulanate elimination is unaltered in this age group. Experience with the 200 mg/5 mL formulation in this age group is limited and, thus, use of the 125 mg/5 mL oral suspension is recommended.
Patients aged 12 weeks (3 months) and older
|
||||||||||||||||||||||
Pediatric patients weighing 40 kg and more should be dosed according to the following adult recommendations: The usual adult dose is 1 Augmentin 500 mg tablet every 12 hours or 1 Augmentin 250 mg tablet every 8 hours. For more severe infections and infections of the respiratory tract, the dose should be 1 Augmentin 875 mg tablet every 12 hours or 1 Augmentin 500 mg tablet every 8 hours. Among adults treated with 875 mg every 12 hours, significantly fewer experienced severe diarrhea or withdrawals with diarrhea vs. adults treated with 500 mg every 8 hours. For detailed adult dosage recommendations, please see complete prescribing information for Augmentin Tablets.
Hepatically impaired patients should be dosed with caution and hepatic function monitored at regular intervals. (See .)
Adults: Adults who have difficulty swallowing may be given the 125 mg/5 mL or 250 mg/5 mL suspension in place of the 500 mg tablet. The 200 mg/5 mL suspension or the 400 mg/5 mL suspension may be used in place of the 875 mg tablet. See dosage recommendations above for children weighing 40 kg or more.
The Augmentin 250 mg tablet and the 250 mg chewable tablet do not contain the same amount of clavulanic acid (as the potassium salt). The Augmentin 250 mg tablet contains 125 mg of clavulanic acid, whereas the 250 mg chewable tablet contains 62.5 mg of clavulanic acid. Therefore, the Augmentin 250 mg tablet and the 250 mg chewable tablet should not be substituted for each other, as they are not interchangeable.
Due to the different amoxicillin to clavulanic acid ratios in the Augmentin 250 mg tablet (250/125) versus the Augmentin 250 mg chewable tablet (250/62.5), the Augmentin 250 mg tablet should not be used until the child weighs at least 40 kg and more.
Prepare a suspension at time of dispensing as follows: Tap bottle until all the powder flows freely. Add approximately 2 / 3 of the total amount of water for reconstitution (see table below) and shake vigorously to suspend powder. Add remainder of the water and again shake vigorously.
|
Each teaspoonful (5 mL) will contain 125 mg amoxicillin and 31.25 mg of clavulanic acid as the potassium salt.
|
Each teaspoonful (5 mL) will contain 200 mg amoxicillin and 28.5 mg of clavulanic acid as the potassium salt.
|
Each teaspoonful (5 mL) will contain 250 mg amoxicillin and 62.5 mg of clavulanic acid as the potassium salt.
|
Each teaspoonful (5 mL) will contain 400 mg amoxicillin and 57.0 mg of clavulanic acid as the potassium salt.
Note: SHAKE ORAL SUSPENSION WELL BEFORE USING.
Administration: Augmentin may be taken without regard to meals; however, absorption of clavulanate potassium is enhanced when Augmentin is administered at the start of a meal. To minimize the potential for gastrointestinal intolerance, Augmentin should be taken at the start of a meal.
AUGMENTIN 125 MG/5 ML FOR ORAL SUSPENSION: Each 5 mL of reconstituted banana-flavored suspension contains 125 mg amoxicillin and 31.25 mg clavulanic acid as the potassium salt.
NDC 0029-6085-39 ...................... 75 mL bottle
NDC 0029-6085-23 .................... 100 mL bottle
NDC 0029-6085-22 .................... 150 mL bottle
AUGMENTIN 200 MG/5 ML FOR ORAL SUSPENSION: Each 5 mL of reconstituted orange-raspberry-flavored suspension contains 200 mg amoxicillin and 28.5 mg clavulanic acid as the potassium salt.
NDC 0029-6087-29 ...................... 50 mL bottle
NDC 0029-6087-39 ...................... 75 mL bottle
NDC 0029-6087-51 .................... 100 mL bottle
AUGMENTIN 250 MG/5 ML FOR ORAL SUSPENSION: Each 5 mL of reconstituted orange-flavored suspension contains 250 mg amoxicillin and 62.5 mg clavulanic acid as the potassium salt.
NDC 0029-6090-39 ...................... 75 mL bottle
NDC 0029-6090-23 .................... 100 mL bottle
NDC 0029-6090-22 .................... 150 mL bottle
AUGMENTIN 400 MG/5 ML FOR ORAL SUSPENSION: Each 5 mL of reconstituted orange-raspberry-flavored suspension contains 400 mg amoxicillin and 57 mg clavulanic acid as the potassium salt.
NDC 0029-6092-29 ...................... 50 mL bottle
NDC 0029-6092-39 ...................... 75 mL bottle
NDC 0029-6092-51 .................... 100 mL bottle
AUGMENTIN 125 MG CHEWABLE TABLETS: Each mottled yellow, round, lemon-lime-flavored tablet, debossed with BMP 189, contains 125 mg amoxicillin as the trihydrate and 31.25 mg clavulanic acid as the potassium salt.
NDC 0029-6073-47 ............ carton of 30 tablets
AUGMENTIN 200 MG CHEWABLE TABLETS: Each mottled pink, round, biconvex, cherry-banana-flavored tablet contains 200 mg amoxicillin as the trihydrate and 28.5 mg clavulanic acid as the potassium salt.
NDC 0029-6071-12 ............ carton of 20 tablets
AUGMENTIN 250 MG CHEWABLE TABLETS: Each mottled yellow, round, lemon-lime-flavored tablet, debossed with BMP 190, contains 250 mg amoxicillin as the trihydrate and 62.5 mg clavulanic acid as the potassium salt.
NDC 0029-6074-47 ............ carton of 30 tablets
AUGMENTIN 400 MG CHEWABLE TABLETS: Each mottled pink, round, biconvex, cherry-banana-flavored tablet contains 400 mg amoxicillin as the trihydrate and 57.0 mg clavulanic acid as the potassium salt.
NDC 0029-6072-12 ............ carton of 20 tablets
AUGMENTIN is also supplied as:
AUGMENTIN 250 MG TABLETS (250 mg amoxicillin/125 mg clavulanic acid):
NDC 0029-6075-27 ...................... bottles of 30
NDC 0029-6075-31 ........ 100 Unit Dose tablets
AUGMENTIN 500 MG TABLETS (500 mg amoxicillin/125 mg clavulanic acid):
NDC 0029-6080-12 ...................... bottles of 20
NDC 0029-6080-31 ........ 100 Unit Dose tablets
AUGMENTIN 875 MG TABLETS (875 mg amoxicillin/125 mg clavulanic acid):
NDC 0029-6086-12 ...................... bottles of 20
NDC 0029-6086-21 ........ 100 Unit Dose tablets
Store tablets and dry powder at or below 25°C (77°F). Dispense in original container. Store reconstituted suspension under refrigeration. Discard unused suspension after 10 days.
 |
In pediatric patients (aged 2 months to 12 years), one U.S./Canadian clinical trial was conducted which compared Augmentin 45/6.4 mg/kg/day (divided q12h) for 10 days versus Augmentin 40/10 mg/kg/day (divided q8h) for 10 days in the treatment of acute otitis media. Only the suspension formulations were used in this trial. A total of 575 patients were enrolled, with an even distribution among the two treatment groups and a comparable number of patients were evaluable (i.e., >/=84%) per treatment group. Strict otitis media-specific criteria were required for eligibility and a strong correlation was found at the end of therapy and follow-up between these criteria and physician assessment of clinical response. The clinical efficacy rates at the end of therapy visit (defined as 2-4 days after the completion of therapy) and at the follow-up visit (defined as 22-28 days post-completion of therapy) were comparable for the two treatment groups, with the following cure rates obtained for the evaluable patients: At end of therapy, 87.2% (n=265) and 82.3% (n=260) for 45 mg/kg/day q12h and 40 mg/kg/day q8h, respectively. At follow-up, 67.1% (n=249) and 68.7% (n=243) for 45 mg/kg/day q12h and 40 mg/kg/day q8h, respectively.
The incidence of diarrhea*** was significantly lower in patients in the q12h treatment group compared to patients who received the q8h regimen (14.3% and 34.3%, respectively). In addition, the number of patients with either severe diarrhea or who were withdrawn with diarrhea was significantly lower in the q12h treatment group (3.1% and 7.6% for the q12h/10 day and q8h/10 day, respectively). In the q12h treatment group, 3 patients (1.0%) were withdrawn with an allergic reaction, while 1 patient (0.3%) in the q8h group was withdrawn for this reason. The number of patients with a candidal infection of the diaper area was 3.8% and 6.2% for the q12h and q8h groups, respectively.
It is not known if the finding of a statistically significant reduction in diarrhea with the oral suspensions dosed q12h, versus suspensions dosed q8h, can be extrapolated to the chewable tablets. The presence of mannitol in the chewable tablets may contribute to a different diarrhea profile. The q12h oral suspensions are sweetened with aspartame only.
*** Diarrhea was defined as either: (a) three or more watery or four or more loose/watery stools in one day; OR (b) two watery stools per day or three loose/watery stools per day for two consecutive days.
Rx only
AG:PL6A