 |
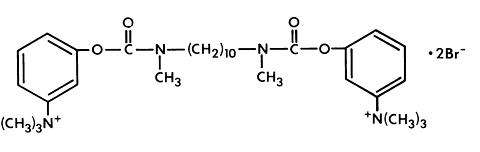
Ophthalmic Solution HUMORSOL* (Demecarium Bromide) is a sterile solution supplied in two dosage strengths: 0.125 percent and 0.25 percent. The inactive ingredients are sodium chloride and water for injection; benzalkonium chloride 1:5000 is added as preservative. Demecarium bromide is a quaternary ammonium compound with a molecular weight of 716.60. Its chemical name is 3,3'-[1,10-decanediylbis [(methylimino)carbonyloxy]] bis [ N,N,N -trimethylbenzenaminium] dibromide. Its empirical formula is C 32 H 52 Br 2 N 4 O 4 and its structural formula is:
 |
*Registered trademark of MERCK & CO., INC.
HUMORSOL is a cholinesterase inhibitor with sustained activity. It acts mainly on true (erythrocyte) cholinesterase. Application of HUMORSOL to the eye produces intense miosis and ciliary muscle contraction due to inhibition of cholinesterase, allowing acetylcholine to accumulate at sites of cholinergic transmission. These effects are accompanied by increased capillary permeability of the ciliary body and iris, increased permeability of the blood-aqueous barrier, and vasodilation. Myopia may be induced or, if present, may be augmented by the increased refractive power of the lens that results from the accommodative effect of the drug. HUMORSOL indirectly produces some of the muscarinic and nicotinic effects of acetylcholine as quantities of the latter accumulate.
Open-angle glaucoma (HUMORSOL should be used in glaucoma only when shorter-acting miotics have proved inadequate.)
Conditions obstructing aqueous outflow, such as synechial formation, that are amenable to miotic therapy
Following iridectomy
Accommodative esotropia (accommodative convergent strabismus)
Hypersensitivity to any component of this product.
Because of the toxicity of cholinesterase inhibitors in general, HUMORSOL is contraindicated in women who are or who may become pregnant. If this drug is used during pregnancy, or if the patient becomes pregnant while taking this drug, the patient should be apprised of the potential hazard to the fetus.
Because miotics may aggravate inflammation, HUMORSOL should not be used in active uveal inflammation and/or glaucoma associated with iridocyclitis.
In patients receiving cholinesterase inhibitors such as HUMORSOL, succinylcholine should be administered with extreme caution before and during general anesthesia.
Because of possible adverse additive effects, HUMORSOL should be administered only with extreme caution to patients with myasthenia gravis who are receiving systemic anticholinesterase therapy; conversely, extreme caution should be exercised in the use of an anticholinesterase drug for the treatment of myasthenia gravis patients who are already undergoing topical therapy with cholinesterase inhibitors.
General
Gonioscopy is recommended prior to medication with HUMORSOL.
HUMORSOL should be used with caution in patients with chronic angle-closure (narrow-angle) glaucoma or in patients with narrow angles, because of the possibility of producing pupillary block and increasing angle blockage.
When an intraocular inflammatory process is present, the intensity and persistence of miosis and ciliary muscle contraction that result from anticholinesterase therapy require abstention from, or cautious use of, HUMORSOL.
Systemic effects are infrequent when HUMORSOL is instilled carefully. Compression of the lacrimal duct for several seconds immediately following instillation minimizes drainage into the nasal chamber with its extensive absorption surface. Wash the hands immediately after instillation.
Discontinue HUMORSOL if salivation, urinary incontinence, diarrhea, profuse sweating, muscle weakness, respiratory difficulties, shock, or cardiac irregularities occur.
Persons receiving cholinesterase inhibitors who are exposed to organophosphate-type insecticides and pesticides (gardeners, organophosphate plant or warehouse workers, farmers, residents of communities which are undergoing insecticide spraying or dusting, etc.) should be warned of the added systemic effects possible from absorption through the respiratory tract or skin. Wearing of respiratory masks, frequent washing, and clothing changes may be advisable.
Anticholinesterase drugs should be used with extreme caution, if at all, in patients with marked vagotonia, bronchial asthma, spastic gastrointesinal disturbances, peptic ulcer, pronounced bradycardia and hypotension, recent myocardial infarction, epilepsy, parkinsonism, and other disorders that may respond adversely to vagotonic effects.
After long-term use of HUMORSOL, dilation of blood vessels and resulting greater permeability increase the possibility of hyphema during ophthalmic surgery. Therefore, this drug should be discontinued before surgery.
Despite observance of all precautions and the use of only the recommended dose, there is some evidence that repeated administration may cause depression of the concentration of cholinesterase in the serum and erythrocytes, with resultant systemic effects.
There have been reports of bacterial keratitis associated with the use of multiple dose containers of topical ophthalmic products. These containers had been inadvertently contaminated by patients who, in most cases, had a concurrent corneal disease or a disruption of the ocular epithelial surface. (See PRECAUTIONS , Information for Patients . )
Information for Patients
Patients should be instructed to avoid allowing the tip of the dispensing container to contact the eye or surrounding structures.
Patients should also be instructed that ocular solutions, if handled improperly, can become contaminated by common bacteria known to cause ocular infections. Serious damage to the eye and subsequent loss of vision may result from using contaminated solutions. (See PRECAUTIONS , General . )
Patients should also be advised that if they develop an intercurrent ocular condition (e.g., trauma, ocular surgery or infection), they should immediately seek their physician' advice concerning the continued use of the present multidose container.
The preservative in HUMORSOL, benzalkonium chloride, may be absorbed by soft contact lenses. Patients wearing soft contact lenses should be instructed to wait at least 15 minutes after instilling HUMORSOL before they insert their lenses.
See regarding possible drug interactions of HUMORSOL with succinylcholine or with other anticholinesterase agents.
Carcinogenesis, Mutagenesis, Impairment of Fertility
Long-term studies in animals have not been performed to evaluate the effects of HUMORSOL on fertility or carcinogenic potential.
Pregnancy
Pregnancy Category X: See CONTRAINDICATIONS .
Nursing Mothers
It is not known whether this drug is excreted in human milk. Because of the potential for serious adverse reactions in nursing infants from HUMORSOL, a decision should be made whether to discontinue nursing or to discontinue the drug, taking into account the importance of the drug to the mother.
Pediatric Use
The occurrence of iris cysts is more frequent in pediatric patients. (See ADVERSE REACTIONS and DOSAGE AND ADMINISTRATION .)
Extreme caution should be exercised in pediatric patients receiving HUMORSOL who may require general anesthesia (see ).
Since HUMORSOL is a potent cholinesterase inhibitor it should be kept out of the reach of children.
Stinging, burning, lacrimation, lid muscle twitching, conjunctival and ciliary redness, brow ache, headache, and induced myopia with visual blurring may occur.
Activation of latent iritis or uveitis may occur.
As with all miotic therapy, retinal detachment has been reported occasionally.
Iris cysts may form, enlarge, and obscure vision. Occurrence is more frequent in children. The iris cyst usually shrinks upon discontinuance of the miotic. Rarely, the cyst may rupture or break free into the aqueous. Frequent examination for this occurrence is advised.
Lens opacities have been reported in patients on miotic therapy. Routine slit-lamp examinations, including the lens, should accompany prolonged use.
Paradoxical increase in intraocular pressure may follow anticholinesterase instillation. This may be alleviated by pupil-dilating medication.
Prolonged use may cause conjunctival thickening and obstruction of nasolacrimal canals.
Systemic effects, which occur rarely, are suggestive of increased cholinergic activity. Such effects may include nausea, vomiting, abdominal cramps, diarrhea, urinary incontinence, salivation, sweating, difficulty in breathing, bradycardia, or cardiac irregularities. Medical management of systemic effects may be indicated (see TREATMENT OF ADVERSE EFFECTS ).
If HUMORSOL is taken systemically by accident, or if systemic effects occur after topical application in the eye or from accidental skin contact, administer atropine sulfate parenterally (intravenously if necessary) in a dose (for adults) of 0.4 to 0.6 mg or more. The recommended dosage of atropine in infants and children up to 12 years of age is 0.01 mg/kg repeated every two hours as needed until the desired effect is obtained, or adverse effects of atropine preclude further usage. The maximum single dose should not exceed 0.4 mg.
The use of much larger doses of atropine in treating anticholinesterase intoxication in adults has been reported in the literature. Initially 2 to 6 mg may be given followed by 2 mg every hour or more often, as long as muscarinic effects continue. The greater possibility of atropinization with large doses, particularly in sensitive individuals, should be borne in mind.
Pralidoxime** chloride has been reported to be useful in treating systemic effects due to cholinesterase inhibitors. However, its use is recommended in addition to and not as substitute for atropine.
A short-acting barbiturate is indicated if convulsions occur that are not entirely relieved by atropine. Barbiturate dosage should be carefully adjusted to avoid central respiratory depression. Marked weakness or paralysis of muscles of respiration should be treated promptly by artificial respiration and maintenance of a clear airway.
The oral LD 50 of HUMORSOL is 2.96 mg/kg in the mouse.
**PROTOPAM® Chloride (Pralidoxime Chloride). Ayerst Laboratories
HUMORSOL is intended solely for topical use in the conjunctival sac.
As HUMORSOL is an extremely potent drug, the physician should thoroughly familiarize himself with its use and the technic of instillation.
The required dose is applied in the conjunctival sac, with the patient supine, care being taken not to touch the cornea with the tip of the OCUMETER* ophthalmic dispenser. The patient or person administering the medication should apply continuous gentle pressure on the lacrimal duct with the index finger for several seconds immediately following instillation of the drops. This is to prevent drainage overflow of solution into the nasal and pharyngeal spaces, which might cause systemic absorption. Wash the hands immediately after administration.
HUMORSOL should not be used more often than directed. Caution is necessary to avoid overdosage.
Initial titration and dosage adjustments with HUMORSOL must be individualized to obtain maximal therapeutic effect. The patient must be closely observed during the initial period. If the response is not adequate within the first 24 hours, other measures should be considered.
Keep frequency of use to a minimum in all patients, but especially in children, to reduce the chance of iris cyst development (see ADVERSE REACTIONS ).
For initial therapy with HUMORSOL (0.125 percent or 0.25 percent) place 1 drop (children) or 1 or 2 drops (adults) in the glaucomatous eye. A decrease in intraocular pressure should occur within a few hours. During this period, keep the patient under supervision and make tonometric examinations at least hourly for 3 or 4 hours to be sure that no immediate rise in pressure occurs (see ADVERSE REACTIONS ).
Duration of effect varies with the individual. The usual dosage can vary from as much as 1 or 2 drops twice a day to as little as 1 or 2 drops twice a week. The 0.125 percent strength used twice a day usually results in smooth control of the physiologic diurnal variation in intraocular pressure. This is probably the preferred dosage for most wide (open) angle glaucoma patients.
Essentially equal visual acuity of both eyes is a prerequisite to the successful treatment of esotropia with HUMORSOL. For initial evaluation it may be used as a diagnostic aid to determine if an accommodative factor exists. This is especially useful preoperatively in young children and in patients with normal hypermetropic refractive errors. One drop is given daily for 2 weeks, then 1 drop every 2 days for 2 to 3 weeks. If the eyes become straighter, an accommodative factor is demonstrated. This technic may supplement or complement standard testing with atropine and trial with glasses for the accommodative factor.
In esotropia uncomplicated by amblyopia or anisometropia, HUMORSOL may be instilled in both eyes, not more than 1 drop at a time every day for 2 to 3 weeks, as too severe a degree of miosis may interfere with vision. Then reduce the dosage to 1 drop every other day for 3 to 4 weeks and reevaluate the patient' status.
HUMORSOL may be continued in a dosage of 1 drop every 2 days to 1 drop twice a week. (The latter dosage may be maintained for several months.) Evaluate the patient' condition every 4 to 12 weeks. If improvement continues, change the schedule to 1 drop once a week and eventually to a trial without medication. However, if after 4 months, control of the condition still requires 1 drop every 2 days, therapy with HUMORSOL should be stopped.
*Registered trademark of MERCK & CO., INC.
Sterile Ophthalmic Solution HUMORSOL is a clear, colorless, aqueous solution and is supplied in a 5 mL white, opaque, plastic OCUMETER ophthalmic dispenser with a controlled-drop tip:
No. 3255--0.125 percent solution.
NDC 0006-3255-03.
No. 3267--0.25 percent solution.
NDC 0006-3267-03.
Protect from freezing and excessive heat.
7414315 Issued September 1996
COPYRIGHT © MERCK & CO., INC., 1987
All rights reserved
 |