 |
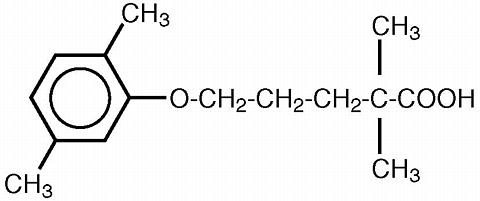
Lopid® (gemfibrozil tablets, USP) is a lipid regulating agent. It is available as tablets for oral administration. Each tablet contains 600 mg gemfibrozil. Each also contains calcium stearate, NF; candelilla wax FCC; microcrystalline cellulose, NF; hydroxypropyl cellulose, NF: hydroxypropyl methylcellulose, USP; methylparaben, NF; Opaspray white; polyethylene glycol, NF; polysorbate 80, NF; propylparaben, NF; colloidal silicon dioxide, NF; pregelatinized starch, NF. The chemical name is 5-(2,5-dimethylphenoxy)-2,2-dimethylpentanoic acid with the following structural formula:
 |
The empirical formula is C 15 H 22 O 3 and the molecular weight is 250.35; the solubility in water and acid is 0.0019% and in dilute base it is greater than 1%. The melting point is 58°-61°C. Gemfibrozil is a white solid which is stable under ordinary conditions.
Lopid (gemfibrozil tablets, USP) is a lipid regulating agent which decreases serum triglycerides and very low density lipoprotein (VLDL) cholesterol, and increases high density lipoprotein (HDL) cholesterol. While modest decreases in total and low density lipoprotein (LDL) cholesterol may be observed with Lopid therapy, treatment of patients with elevated triglycerides due to Type IV hyperlipoproteinemia often results in a rise in LDL-cholesterol. LDL-cholesterol levels in Type IIb patients with elevations of both serum LDL-cholesterol and triglycerides are, in general, minimally affected by Lopid treatment; however, Lopid usually raises HDL-cholesterol significantly in this group. Lopid increases levels of high density lipoprotein (HDL) subfractions HDL 2 and HDL 3 , as well as apolipoproteins AI and AII. Epidemiological studies have shown that both low HDL-cholesterol and high LDL-cholesterol are independent risk factors for coronary heart disease.
In the primary prevention component of the Helsinki Heart Study (refs. 1,2), in which 4081 male patients between the ages of 40 and 55 were studied in a randomized, double-blind, placebo-controlled fashion, Lopid therapy was associated with significant reductions in total plasma triglycerides and a significant increase in high density lipoprotein cholesterol. Moderate reductions in total plasma cholesterol and low density lipoprotein cholesterol were observed for the Lopid treatment group as a whole, but the lipid response was heterogeneous, especially among different Fredrickson types. The study involved subjects with serum non-HDL-cholesterol of over 200 mg/dL and no previous history of coronary heart disease. Over the 5-year study period, the Lopid group experienced a 1.4% absolute (34% relative) reduction in the rate of serious coronary events (sudden cardiac deaths plus fatal and nonfatal myocardial infarctions) compared to placebo, p = 0.04 (see Table I). There was a 37% relative reduction in the rate of nonfatal myocardial infarction compared to placebo, equivalent to a treatment-related difference of 13.1 events per thousand persons. Deaths from any cause during the double-blind portion of the study totaled 44 (2.2%) in the Lopid randomization group and 43 (2.1%) in the placebo group.
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Among Fredrickson types, during the 5-year double-blind portion of the primary prevention component of the Helsinki Heart Study, the greatest reduction in the incidence of serious coronary events occurred in Type IIb patients who had elevations of both LDL-cholesterol and total plasma triglycerides. This subgroup of Type IIb gemfibrozil group patients had a lower mean HDL-cholesterol level at baseline than the Type IIa subgroup that had elevations of LDL-cholesterol and normal plasma triglycerides. The mean increase in HDL-cholesterol among the Type IIb patients in this study was 12.6% compared to placebo. The mean change in LDL-cholesterol among Type IIb patients was -4.1% with Lopid compared to a rise of 3.9% in the placebo subgroup. The Type IIb subjects in the Helsinki Heart Study had 26 fewer coronary events per thousand persons over 5 years in the gemfibrozil group compared to placebo. The difference in coronary events was substantially greater between Lopid and placebo for that subgroup of patients with the triad of LDL-cholesterol >175 mg/dL (>4.5 mmol), triglycerides >200 mg/dL (>2.2 mmol), and HDL-cholesterol <35 mg/dL (<0.90 mmol) (see Table I).
Further information is available from a 3.5 year (8.5 year cumulative) follow-up of all subjects who had participated in the Helsinki Heart Study. At the completion of the Helsinki Heart study, subjects could choose to start, stop, or continue to receive Lopid; without knowledge of their own lipid values or double-blind treatment, 60% of patients originally randomized to placebo began therapy with Lopid and 60% of patients originally randomized to Lopid continued medication. After approximately 6.5 years following randomization, all patients were informed of their original treatment group and lipid values during the 5 years of the double-blind treatment. After further elective changes in Lopid treatment status, 61% of patients in the group originally randomized to Lopid were taking drug; in the group originally randomized to placebo, 65% were taking Lopid. The event rate per 1000 occurring during the open-label follow-up period is detailed in Table II.
|
||||||||||||||||||||||||||||
Cumulative mortality through 8.5 years showed a 20% relative excess of deaths in the group originally randomized to Lopid versus the originally randomized placebo group and a 20% relative decrease in cardiac events in the group originally randomized to Lopid versus the originally randomized placebo group (see Table III). This analysis of the originally randomized "intent-to-treat" population neglects the possible complicating effects of treatment switching during the open-label phase. Adjustment of hazard ratios taking into account open-label treatment status from years 6.5 to 8.5 could change the reported hazard ratios for mortality toward unity.
|
|||||||||||||||||||||||||||||||||||||||||||||
It is not clear to what extent the findings of the primary prevention component of the Helsinki Heart Study can be extrapolated to other segments of the dyslipidemic population not studied (such as women, younger or older males, or those with lipid abnormalities limited solely to HDL-cholesterol) or to other lipid-altering drugs.
The secondary prevention component of the Helsinki Heart Study was conducted over 5 years in parallel and at the same centers in Finland in 628 middle-aged males excluded from the primary prevention component of the Helsinki Heart Study because of a history of angina, myocardial infarction or unexplained ECG changes (ref. 3). The primary efficacy endpoint of the study was cardiac events (the sum of fatal and non-fatal myocardial infarctions and sudden cardiac deaths). The hazard ratio (Lopid:placebo) for cardiac events was 1.47 (95% confidence limits 0.88-2.48, p = 0.14). Of the 35 patients in the Lopid group who experienced cardiac events, 12 patients suffered events after discontinuation from the study. Of the 24 patients in the placebo group with cardiac events, 4 patients suffered events after discontinuation from the study. There were 17 cardiac deaths in the Lopid group and 8 in the placebo group (hazard ratio 2.18; 95% confidence limits 0.94-5.05, p = 0.06). Ten of these deaths in the Lopid group and 3 in the placebo group occurred after discontinuation from therapy. In this study of patients with known or suspected coronary heart disease, no benefit from Lopid treatment was observed in reducing cardiac events or cardiac deaths. Thus, Lopid has shown benefit only in selected dyslipidemic patients without suspected or established coronary heart disease. Even in patients with coronary heart disease and the triad of elevated LDL-cholesterol, elevated triglycerides, plus low HDL-cholesterol, the possible effect of Lopid on coronary events has not been adequately studied.
No efficacy in the patients with established coronary heart disease was observed during the Coronary Drug Project with the chemically and pharmacologically related drug, clofibrate. The Coronary Drug Project was a 6-year randomized, double-blind study involving 1000 clofibrate, 1000 nicotinic acid, and 3000 placebo patients with known coronary heart disease. A clinically and statistically significant reduction in myocardial infarctions was seen in the concurrent nicotinic acid group compared to placebo; no reduction was seen with clofibrate.
The mechanism of action of gemfibrozil has not been definitely established. In man, Lopid has been shown to inhibit peripheral lipolysis and to decrease the hepatic extraction of free fatty acids, thus reducing hepatic triglyceride production. Lopid inhibits synthesis and increases clearance of VLDL carrier apolipoprotein B, leading to a decrease in VLDL production.
Animal studies suggest that gemfibrozil may, in addition to elevating HDL-cholesterol, reduce incorporation of long-chain fatty acids into newly formed triglycerides, accelerate turnover and removal of cholesterol from the liver, and increase excretion of cholesterol in the feces. Lopid is well absorbed from the gastrointestinal tract after oral administration. Peak plasma levels occur in 1 to 2 hours with a plasma half-life of 1.5 hours following multiple doses.
Gemfibrozil is completely absorbed after oral administration of Lopid tablets, reaching peak plasma concentrations 1 to 2 hours after dosing. Gemfibrozil pharmacokinetics are affected by the timing of meals relative to time of dosing. In one study (ref. 4), both the rate and extent of absorption of the drug were significantly increased when administered 0.5 hours before meals. Average AUC was reduced by 14-44% when Lopid was administered after meals compared to 0.5 hours before meals. In a subsequent study (ref. 4), rate of absorption of Lopid was maximum when administered 0.5 hours before meals with the Cmax 50-60% greater than when given either with meals or fasting. In this study, there were no significant effects on AUC of timing of dose relative to meals (see DOSAGE AND ADMINISTRATION ).
Lopid mainly undergoes oxidation of a ring methyl group to successively form a hydroxymethyl and a carboxyl metabolite. Approximately seventy percent of the administered human dose is excreted in the urine, mostly as the glucuronide conjugate, with less than 2% excreted as unchanged gemfibrozil. Six percent of the dose is accounted for in the feces. Gemfibrozil is highly bound to plasma proteins and there is potential for displacement interactions with other drugs (see PRECAUTIONS ).
Lopid (gemfibrozil tablets, USP) is indicated as adjunctive therapy to diet for:
In a subgroup analysis of patients in the Helsinki Heart Study with above-median HDL-cholesterol values at baseline (greater than 46.4 mg/dL), the incidence of serious coronary events was similar for gemfibrozil and placebo subgroups (see Table I).
The initial treatment for dyslipidemia is dietary therapy specific for the type of lipoprotein abnormality. Excess body weight and excess alcohol intake may be important factors in hypertriglyceridemia and should be managed prior to any drug therapy. Physical exercise can be an important ancillary measure, and has been associated with rises in HDL-cholesterol. Diseases contributory to hyperlipidemia such as hypothyroidism or diabetes mellitus should be looked for and adequately treated. Estrogen therapy is sometimes associated with massive rises in plasma triglycerides, especially in subjects with familial hypertriglyceridemia. In such cases, discontinuation of estrogen therapy may obviate the need for specific drug therapy of hypertriglyceridemia. The use of drugs should be considered only when reasonable attempts have been made to obtain satisfactory results with nondrug methods. If the decision is made to use drugs, the patient should be instructed that this does not reduce the importance of adhering to diet
1. Initial Therapy-- Laboratory studies should be done to ascertain that the lipid levels are consistently abnormal. Before instituting Lopid therapy, every attempt should be made to control serum lipids with appropriate diet, exercise, weight loss in obese patients, and control of any medical problems such as diabetes mellitus and hypothryroidism that are contributing to the lipid abnormalities.
2. Continued Therapy-- Periodic determination of serum lipids should be obtained, and the drug withdrawn if lipid response is inadequate after 3 months of therapy.
3. Drug Interactions--(A) HMG-CoA reductase inhibitors: Rhabdomyolysis has occurred with combined gemfibrozil and lovastatin therapy. It may be seen as early as 3 weeks after initiation of combined therapy or after several months. In most subjects who have had an unsatisfactory lipid response to either drug alone, the possible benefit of combined therapy with lovastatin (or other HMG-CoA reductase inhibitors) and gemfibrozil does not outweigh the risks of severe myopathy, rhabdomyolysis, and acute renal failure. There is no assurance that periodic monitoring of creatine kinase will prevent the occurrence of severe myopathy and kidney damage.
(B) Anticoagulants: CAUTION SHOULD BE EXERCISED WHEN ANTICOAGULANTS ARE GIVEN IN CONJUNCTION WITH LOPID. THE DOSAGE OF THE ANTICOAGULANT SHOULD BE REDUCED TO MAINTAIN THE PROTHROMBIN TIME AT THE DESIRED LEVEL TO PREVENT BLEEDING COMPLICATIONS. FREQUENT PROTHROMBIN DETERMINATIONS ARE ADVISABLE UNTIL IT HAS BEEN DEFINITELY DETERMINED THAT THE PROTHROMBIN LEVEL HAS STABILIZED.
4. Carcinogenesis, Mutagenesis, Impairment of Fertility-- Long-term studies have been conducted in rats at 0.2 and 1.3 times the human exposure (based on AUC). Based on two-week toxicokinetic studies, exposure (AUC) of the dose groups was estimated to be 0.2 and 1.3 times the human exposure. The incidence of benign liver nodules and liver carcinomas was significantly increased in high dose male rats. The incidence of liver carcinomas increased also in low dose males, but this increase was not statistically significant (p=0.1). Male rats had a dose-related and statistically significant increase of benign Leydig cell tumors. The higher dose female rats had a significant increase in the combined incidence of benign and malignant liver neoplasms.
Long-term studies have been conducted in mice at 0.1 and 0.7 times the human exposure (based on AUC). There were no statistically significant differences from controls in the incidence of liver tumors, but the doses tested were lower than those shown to be carcinogenic with other fibrates.
Electron microscopy studies have demonstrated a florid hepatic peroxisome proliferation following Lopid administration to the male rat. An adequate study to test for peroxisome proliferation has not been done in humans, but changes in peroxisome morphology have been observed. Peroxisome proliferation has been shown to occur in humans with either of two other drugs of the fibrate class when liver biopsies were compared before and after treatment in the same individual.
Administration of approximately 0.6 and 2 times the human dose (based on surface area) to male rats for 10 weeks resulted in a dose-related decrease of fertility. Subsequent studies demonstrated that this effect was reversed after a drug-free period of about eight weeks, and it was not transmitted to the offspring.
5. Pregnancy Category C-- Lopid has been shown to produce adverse effects in rats and rabbits at doses between 0.5 and 3 times the human dose (based on surface area). There are no adequate and well-controlled studies in pregnant women. Lopid should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus.
Administration of Lopid to female rats at 0.6 and 2 times the human dose (based on surface area) before and throughout gestation caused a dose-related decrease in conception rate and, at the high dose, an increase in stillborns and a slight reduction in pup weight during lactation. There were also dose-related increased skeletal variations. Anophthalmia occurred, but rarely.
Administration of 0.6 and 2 times the human dose (based on surface area) of Lopid to female rats from gestation day 15 through weaning caused dose-related decreases in birth weight and suppressions of pup growth during lactation.
Administration of 1 and 3 times the human dose (based on surface area) of Lopid to female rabbits during organogenesis caused a dose-related decrease in litter size and, at the high dose, an increased incidence of parietal bone variations.
6. Nursing Mothers-- It is not known whether this drug is excreted in human milk. Because many drugs are excreted in human milk and because of the potential for tumorigenicity shown for Lopid in animal studies, a decision should be made whether to discontinue nursing or to discontinue the drug, taking into account the importance of the drug to the mother.
7. Hematologic Changes-- Mild hemoglobin, hematocrit and white blood cell decreases have been observed in occasional patients following initiation of Lopid therapy. However, these levels stabilize during long-term administration. Rarely, severe anemia, leukopenia, thrombocytopenia, and bone marrow hypoplasia have been reported. Therefore, periodic blood counts are recommended during the first 12 months of Lopid administration.
8. Liver Function-- Abnormal liver function tests have been observed occasionally during Lopid administration, including elevations of AST (SGOT), ALT (SGPT), LDH, bilirubin, and alkaline phosphatase. These are usually reversible when Lopid is discontinued. Therefore periodic liver function studies are recommended and Lopid therapy should be terminated if abnormalities persist.
9. Kidney Function-- There have been reports of worsening renal insufficiency upon the addition of Lopid therapy in individuals with baseline plasma creatinine >2.0 mg/dL. In such patients, the use of alternative therapy should be considered against the risks and benefits of a lower dose of Lopid.
10. Use in Pediatric Patients-- Safety and efficacy in pediatric patients have not been established.
In the double-blind controlled phase of the primary prevention component of the Helsinki Heart Study, 2046 patients received Lopid for up to 5 years. In that study, the following adverse reactions were statistically more frequent in subjects in the Lopid group:
|
||||||||||||||||||||||||
Adverse events reported by more than 1% of subjects, but without a significant difference between groups:
|
Gallbladder surgery was performed in 0.9% of Lopid and 0.5% of placebo subjects in the primary prevention component, a 64% excess, which is not statistically different from the excess of gallbladder surgery observed in the clofibrate compared to the placebo group of the WHO study. Gallbladder surgery was also performed more frequently in the Lopid group compared to placebo (1.9% vs 0.3%, p = 0.07) in the secondary prevention component. A statistically significant increase in appendectomy in the gemfibrozil group was seen also in the secondary prevention component (6 on gemfibrozil vs 0 on placebo, p = 0.014).
Nervous system and special senses adverse reactions were more common in the Lopid group. These included hypesthesia, paresthesias, and taste perversion. Other adverse reactions that were more common among Lopid treatment group subjects but where a causal relationship was not established include cataracts, peripheral vascular disease, and intracerebral hemorrhage.
From other studies it seems probable that Lopid is causally related to the occurrence of MUSCULOSKELETAL SYMPTOMS (see ), and to ABNORMAL LIVER FUNCTION TESTS and HEMATOLOGIC CHANGES (see PRECAUTIONS).
Reports of viral and bacterial infections (common cold, cough, urinary tract infections) were more common in gemfibrozil treated patients in other controlled clinical trials of 805 patients. Additional adverse reactions that have been reported for gemfibrozil are listed below by system. These are categorized according to whether a causal relationship to treatment with Lopid is probable or not established:
|
The recommended dose for adults is 1200 mg administered in two divided doses 30 minutes before the morning and evening meal (see ).
There have been reported cases of overdosage with Lopid. In one case a 7 year old child recovered after ingesting up to 9 grams of Lopid. Symptomatic supportive measures should be taken should an overdose occur.
Lopid (Tablet 737), white, elliptical, film-coated, scored tablets, each containing 600 mg gemfibrozil, are available as follows:
N 0071-0737-20: Bottles of 60
N 0071-0737-30: Bottles of 500
Parcode No. 737
Storage: Store at controlled room temperature 20°-25° C (68°-77° F) [see USP]. Protect from light and humidity.
 |
Rx only
Revised May 1999
Manufactured by:
Parke Davis Pharmaceuticals, Ltd.
Vega Baja, PR 00694
Distributed by:
PARKE-DAVIS
Div of Warner-Lambert Co
Morris Plains, NJ 07950 USA
©1997-'99, PDPL
0737G303