 |
*This product contains dry natural rubber
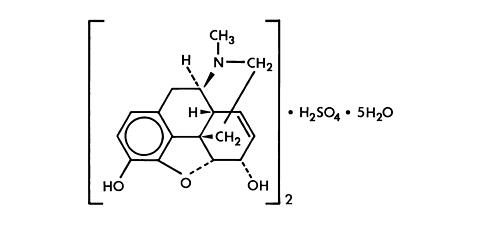
Chemically, morphine sulfate is 7,8 didehydro-4,5 (alpha)-epoxy-17-methylmorphinan-3,6 (alpha)-diol sulfate (2:1) (salt) pentahydrate and has the following structural formula:
 |
Each 5 mL of MSIR Oral Solution contains:
Morphine Sulfate .......... 10 or 20 mg
Inactive Ingredients: Edetate disodium, FD&C Red. No. 40, Glycerin, Invert Sugar, Sodium benzoate, Sodium chloride, Sucrose, Artificial & Natural Flavors, and other ingredients.
Each 1 mL of MSIR Oral Solution Concentrate contains:
Morphine Sulfate .......... 20 mg
Inactive Ingredients: Edetate disodium, Sodium benzoate, and other ingredients.
Each MSIR Tablet for oral administration contains:
Morphine Sulfate .......... 15 or 30 mg
Inactive Ingredients: Croscarmellose sodium, Lactose, Magnesium stearate, Microcrystalline cellulose, and Talc.
Each MSIR Capsule for oral administration contains:
Morphine Sulfate .......... 15 or 30 mg
Inactive Ingredients: FD&C Blue No. 1, FD&C Blue No. 2, FD&C Red No. 40, FD&C Yellow No. 6, Gelatin, Hydroxypropyl methylcellulose, Lactose, Polyethylene glycol, Polysorbate 80, Polyvinylpyrrolidone, Starch, Sucrose, Titanium dioxide, and other ingredients. In addition, the 30 mg capsule contains Black iron oxide and D&C Red No. 28.
MSIR Solutions, Tablets and Capsules containing morphine sulfate are for oral administration and are conventional immediate release products. Only about 40% of the administered dose reaches the central compartment because of pre-systemic elimination (i.e., metabolism in the gut wall and liver).
Once absorbed, morphine is distributed to skeletal muscle, kidneys, liver, intestinal tract, lungs, spleen and brain. Morphine also crosses the placental membranes and has been found in breast milk.
Although a small fraction (less than 5%) of morphine is demethylated, for all practical purposes, virtually all morphine is converted to glucuronide metabolites; among these, morphine-3-glucuronide is present in the highest plasma concentration following oral administration.
The glucuronide system has a very high capacity and is not easily saturated even in disease. Therefore, rate of delivery of morphine to the gut and liver should not influence the total and, probably, the relative quantities of the various metabolites formed. Moreover, even if rate affected the relative amounts of each metabolite formed, it should be unimportant clinically because morphine' metabolites are ordinarily inactive.
The following pharmacokinetic parameters show considerable intersubject variation but are representative of average values reported in the literature. The volume of distribution (Vd) for morphine is 4 liters per kilogram, and its terminal elimination half-life is approximately 2 to 4 hours. Following the administration of conventional oral morphine products, approximately fifty percent of the morphine that will reach the central compartment intact, reaches it within 30 minutes.
Variation in the physical/mechanical properties of a formulation of an oral morphine drug product can affect both its absolute bioavailability and its absorption rate constant (k a ). The basic pharmacokinetic parameters (e.g., volume of distribution [Vd], elimination rate constant [k e ], clearance [Cl]) are fundamental properties of morphine in the organism. However, in chronic use, the possibility that shifts in metabolite to parent drug ratios may occur cannot be excluded.
When immediate-release oral morphine is given on a fixed dosing regimen, steady state is achieved in about a day.
For a given dose and dosing interval, the AUC and average blood concentration of morphine at steady state (Css) will be independent of the specific type of oral formulation administered so long as the formulations have the same absolute bioavailability. The absorption rate of a formulation will, however, affect the maximum (Cmax) and minimum (Cmin) blood levels and the times of their occurrence.
While there is no predictable relationship between morphine blood levels and analgesic response, effective analgesia will not occur below some minimum blood level in a given patient. The minimum effective blood level for analgesia will vary among patients, especially among patients who have been previously treated with potent mu (µ) agonist opioids. Similarly, there is no predictable relationship between blood morphine concentration and untoward clinical responses; again, however, higher concentrations are more likely to be toxic than lower ones.
The elimination of morphine occurs primarily as renal excretion of 3-morphine glucuronide. A small amount of the glucuronide conjugate is excreted in the bile, and there is some minor enterohepatic recycling.
The elimination half-life of morphine is reported to vary between 2 and 4 hours. Thus, steady-state is probably achieved on most regimens within a day. Because morphine is primarily metabolized to inactive metabolites, the effects of renal disease on morphine' elimination are not likely to be pronounced. However, as with any drug, caution should be taken to guard against unanticipated accumulation if renal and/or hepatic function is seriously impaired.
Individual differences in the metabolism of morphine suggest that MSIR Oral Solutions, Tablets and Capsules be dosed conservatively according to the dosing initiation and titration recommendations in the Dosage and Administration section.
The effects described below are common to all morphine-containing products.
The principal actions of therapeutic value of morphine are analgesia and sedation (i.e., sleepiness and anxiolysis).
The precise mechanism of analgesic action is unknown. However, specific CNS opiate receptors and endogenous compounds with morphine-like activity have been identified throughout the brain and spinal cord and are likely to play a role in the expression of analgesic effects.
Morphine produces respiratory depression by direct action on brain stem respiratory centers. The mechanism of respiratory depression involves a reduction in the responsiveness of the brain stem respiratory centers to increases in carbon dioxide tension, and to electrical stimulation.
Morphine depresses the cough reflex by direct effect on the cough center in the medulla. Antitussive effects may occur with doses lower than those usually required for analgesia.
Morphine causes miosis, even in total darkness. Pinpoint pupils are a sign of narcotic overdose but are not pathognomonic (e.g., pontine lesions of hemorrhagic or ischemic origins may produce similar findings). Marked mydriasis rather than miosis may be seen with worsening hypoxia.
Gastric, biliary and pancreatic secretions are decreased by morphine. Morphine causes a reduction in motility associated with an increase in tone in the antrum of the stomach and duodenum. Digestion of food in the small intestine is delayed and propulsive contractions are decreased. In addition, propulsive peristaltic waves in the colon are decreased, while tone is increased to the point of spasm. The end result is constipation. Morphine can cause a marked increase in biliary tract pressure as a result of spasm of the sphincter of Oddi.
Morphine produces peripheral vasodilation which may result in orthostatic hypotension. Release of histamine can occur and may contribute to narcotic-induced hypotension. Manifestations of histamine release and/or peripheralvasodilation may include pruritus, flushing, red eyes and sweating.
MSIR Oral Solutions, Tablets and Capsules are indicated for the relief of moderate to severe pain.
MSIR Oral Solutions, Tablets and Capsules are contraindicated in patients with known hypersensitivity to the drug, in patients with respiratory depression in the absence of resuscitative equipment, and in patients with acute or severe bronchial asthma.
MSIR Oral Solutions, Tablets and Capsules are contraindicated in any patient who has or is suspected of having a paralytic ileus.
Respiratory depression is the chief hazard of all morphine preparations. Respiratory depression occurs most frequently in elderly and debilitated patients, and those suffering from conditions accompanied by hypoxia or hypercapnia when even moderate therapeutic doses may dangerously decrease pulmonary ventilation.
Morphine should be used with extreme caution in patients with chronic obstructive pulmonary disease or cor pulmonale, and in patients having a substantially decreased respiratory reserve, hypoxia, hypercapnia, or preexisting respiratory depression. In such patients, even usual therapeutic doses of morphine may decrease respiratory drive while simultaneously increasing airway resistance to the point of apnea.
The respiratory depressant effects of morphine with carbon dioxide retention and secondary elevation of cerebrospinal fluid pressure may be markedly exaggerated in the presence of head injury, other intracranial lesions, or preexisting increase in intracranial pressure. Morphine produces effects which may obscure neurologic signs of further increase in pressure in patients with head injuries.
MSIR Oral Solutions, Tablets and Capsules, like all opioid analgesics, may cause severe hypotension in an individual whose ability to maintain his blood pressure has already been compromised by a depleted blood volume, or a concurrent administration of drugs such as phenothiazines, or general anesthetics. (See also: PRECAUTIONS: Drug Interactions.) MSIR Oral Solutions, Tablets and Capsules may produce orthostatic hypotension in ambulatory patients.
MSIR Oral Solutions, Tablets and Capsules, like all opioid analgesics, should be administered with caution to patients in circulatory shock, since vasodilation produced by the drug may further reduce cardiac output and blood pressure.
MSIR Oral Solutions, Tablets and Capsules, like all opioid analgesics, should be used with great caution and in reduced dosage in patients who are concurrently receiving other central nervous system depressants including sedatives or hypnotics, general anesthetics, phenothiazines, other tranquilizers and alcohol, because respiratory depression, hypotension and profound sedation or coma may result.
From a theoretical perspective, agonist/antagonist analgesics (i.e., pentazocine, nalbuphine, butorphanol and buprenorphine) should NOT be administered to a patient who has received or is receiving a course of therapy with a pure agonist opioid analgesic. In these patients, mixed agonist-antagonist analgesics may reduce the analgesic effect or may precipitate withdrawal symptoms.
Morphine can produce drug dependence and has a potential for being abused. Tolerance and psychological and physical dependence may develop upon repeated administration. Physical dependence, however, is not of paramount importance in the management of terminally ill patients or any patient in severe pain. Abrupt cessation or a sudden reduction in dose after prolonged use may result in withdrawal symptoms. After prolonged exposure to opioid analgesics, if withdrawal is necessary, it must be undertaken gradually. (See DRUG ABUSE AND DEPENDENCE .)
Infants born to mothers physically dependent on opioid analgesics may also be physically dependent and exhibit respiratory depression and withdrawal symptoms. (See DRUG ABUSE AND DEPENDENCE .)
PRECAUTIONS (See also: CLINICAL PHARMACOLOGY)
MSIR Oral Solutions, Tablets and Capsules are intended for use in patients who require a potent opioid analgesic for relief of moderate to severe pain.
Selection of patients for treatment with MSIR Oral Solutions, Tablets and Capsules should be governed by the same principles that apply to the use of morphine and other potent opioid analgesics. Specifically, the increased risks associated with its use in the following populations should be considered: the elderly or debilitated and those with severe impairment of hepatic, pulmonary or renal function; myxedema or hypothyroidism; adrenocortical insufficiency (e.g., Addison' Disease); CNS depression or coma; toxic psychoses; prostatic hypertrophy or urethral stricture; acute alcoholism; delirium tremens; kyphoscoliosis or inability to swallow.
The administration of morphine, like all opioid analgesics, may obscure the diagnosis or clinical course in patients with acute abdominal conditions.
Morphine may aggravate preexisting convulsions in patients with convulsive disorders.
Morphine should be used with caution in patients about to undergo surgery of the biliary tract, since it may cause spasm of the sphincter of Oddi. Similarly, morphine should be used with caution in patients with acute pancreatitis secondary to biliary tract disease.
If clinically advisable, patients receiving MSIR Oral Solutions, Tablets and Capsules should be given the following instructions by the physician.
1.Morphine may produce physical and/or psychological dependence. For this reason, the dose of the drug should not be adjusted without consulting a physician.
2.Morphine may impair mental and/or physical ability required for the performance of potentially hazardous tasks (e.g., driving, operating machinery).
3.Morphine should not be taken with alcohol or other CNS depressants (sleep aids, tranquilizers) because additive effects including CNS depression may occur. A physician should be consulted if other prescription medications are currently being used or are prescribed for future use.
4.For women of childbearing potential who become or are planning to become pregnant, a physician should be consulted regarding analgesics and other drug use.
Drug Interactions (See also )
The concomitant use of other central nervous system depressants including sedatives or hypnotics, general anesthetics, phenothiazines, tranquilizers and alcohol may produce additive depressant effects. Respiratory depression, hypotension and profound sedation or coma may occur. When such combined therapy is contemplated, the dose of one or both agents should be reduced. Opioid analgesics, including MSIR Oral Solutions, Tablets and Capsules, may enhance the neuromuscular blocking action of skeletal muscle relaxants and produce an increased degree of respiratory depression.
Studies of morphine sulfate in animals to evaluate the drug' carcinogenic and mutagenic potential or the effect on fertility have not been conducted.
Teratogenic effects--CATEGORY C: Adequate animal studies on reproduction have not been performed to determine whether morphine affects fertility in males or females. There are no well-controlled studies in women, but marketing experience does not include any evidence of adverse effects on the fetus following routine (short-term) clinical use of morphine sulfate products. Although there is no clearly defined risk, such experience cannot exclude the possibility of infrequent or subtle damage to the human fetus. MSIR Oral Solutions, Tablets and Capsules should be used in pregnant women only when clearly needed. (See also: PRECAUTIONS: Labor and Delivery, and DRUG ABUSE AND DEPENDENCE.)
Nonteratogenic effects: Infants born from mothers who have been taking morphine chronically may exhibit withdrawal symptoms.
MSIR Oral Solutions, Tablets and Capsules are not recommended for use in women during and immediately prior to labor. Occasionally, opioid analgesics may prolong labor through actions which temporarily reduce the strength, duration and frequency of uterine contractions. However, this effect is not consistent and may be offset by an increased rate of cervical dilatation which tends to shorten labor.
Neonates whose mothers received opioid analgesics during labor should be observed closely for signs of respiratory depression. A specific narcotic antagonist, naloxone, should be available for reversal of narcotic-induced respiratory depression in the neonate.
Low levels of morphine have been detected in human milk. Withdrawal symptoms can occur in breast-feeding infants when maternal administration of morphine sulfate is stopped. Nursing should not be undertaken while a patient is receiving MSIR Oral Solutions, Tablets and Capsules since morphine may be excreted in the milk.
MSIR Oral Solutions, Tablets and Capsules have not been evaluated systematically in children.
The adverse reactions caused by morphine are essentially the same as those observed with other opioid analgesics. They include the following major hazards: respiratory depression, apnea, and to a lesser degree, circulatory depression; respiratory arrest, shock, and cardiac arrest.
Constipation, lightheadedness, dizziness, sedation, nausea, vomiting, sweating, dysphoria and euphoria.
Some of these effects seem to be more prominent in ambulatory patients and in those not experiencing severe pain. Some adverse reactions in ambulatory patients may be alleviated if the patient lies down.
Central Nervous System: Weakness, headache, agitation, tremor, uncoordinated muscle movements, seizure, alterations of mood (nervousness, apprehension, depression, floating feelings), dreams, muscle rigidity, transient hallucinations and disorientation, visual disturbances, insomnia and increased intracranial pressure.
Gastrointestinal: Dry mouth, biliary tract spasm, laryngospasm, anorexia, diarrhea, cramps and taste alterations.
Cardiovascular: Flushing of the face, chills, tachycardia, bradycardia, palpitation, faintness, syncope, hypotension and hypertension.
Genitourinary: Urinary retention or hesitance, reduced libido, and/or potency.
Dermatologic: Pruritus, urticaria, other skin rashes, edema and diaphoresis.
Other: Antidiuretic effect, paresthesia, muscle tremor, blurred vision, nystagmus, diplopia and miosis.
Opioid analgesics may cause psychological and physical dependence. (See .) Physical dependence results in withdrawal symptoms in patients who abruptly discontinue the drug or may be precipitated through the administration of drugs with narcotic antagonist activity, e.g., naloxone or mixed agonist/antagonist analgesics (pentazocine, etc.: see also OVERDOSE ). Physical dependence usually does not occur to a clinically significant degree until after several weeks of continued narcotic usage. Tolerance, in which increasingly large doses are required in order to produce the same degree of analgesia, is initially manifested by a shortened duration of analgesic effect, and, subsequently, by decreases in the intensity of analgesia.
In chronic-pain patients and in narcotic-tolerant cancer patients, the administration of MSIR Oral Solutions, Tablets and Capsules should be guided by the degree of tolerance manifested. Physical dependence, per se, is not ordinarily a concern when one is dealing with opioid-tolerant patients whose pain and suffering is associated with an irreversible illness.
If MSIR Oral Solutions, Tablets and Capsules are abruptly discontinued, a moderate to severe abstinence syndrome may occur. The opioid agonist abstinence syndrome is characterized by some or all of the following: restlessness, lacrimation, rhinorrhea, yawning, perspiration, cutis anserina, restless sleep known as the "yen" and mydriasis during the first 24 hours. These symptoms often increase in severity and over the next 72 hours may be accompanied by increasing irritability, anxiety, weakness, twitching and spasms of muscles; kicking movements; severe backache, abdominal and leg pains; abdominal and muscle cramps; hot and cold flashes; insomnia; nausea, anorexia, vomiting, intestinal spasm, diarrhea; coryza and repetitive sneezing; and increase in body temperature, blood pressure, respiratory rate and heart rate. Because of excessive loss of fluids through sweating, vomiting and diarrhea, there is usually marked weight loss, dehydration, ketosis, and disturbances in acid-base balance. Cardiovascular collapse can occur. Without treatment, most observable symptoms disappear in 5-14 days; however, there appears to be a phase of secondary or chronic abstinence which may last for 2-6 months, characterized by insomnia, irritability, and muscular aches.
If treatment of physical dependence on MSIR Oral Solutions, Tablets and Capsules is necessary, the patient may be detoxified by gradual reduction of the dosage. Gastrointestinal disturbances or dehydration should be treated accordingly.
Acute overdosage with morphine is manifested by respiratory depression, somnolence progressing to stupor or coma, skeletal muscle flaccidity, cold and clammy skin, constricted pupils, and, sometimes, bradycardia and hypotension.
In the treatment of overdosage, primary attention should be given to the re-establishment of a patent airway and institution of assisted or controlled ventilation. The pure opioid antagonist, naloxone, is a specific antidote against respiratory depression which results from opioid overdose. Naloxone (usually 0.4 to 2.0 mg) should be administered intravenously; however, because its duration of action is relatively short, the patient must be carefully monitored until spontaneous respiration is reliably reestablished. If the response to naloxone is suboptimal or not sustained, additional naloxone may be re-administered, as needed, or given by continuous infusion to maintain alertness and respiratory function; however, there is no information available about the cumulative dose of naloxone that may be safely administered.
Naloxone should not be administered in the absence of clinically significant respiratory or circulatory depression secondary to morphine overdose. Naloxone should be administered cautiously to persons who are known or suspected to be physically dependent on morphine. In such cases, an abrupt or complete reversal of narcotic effects may precipitate an acute abstinence syndrome.
Note: In an individual physically dependent on opioids, administration of the usual dose of the antagonist will precipitate an acute withdrawal syndrome. The severity of the withdrawal syndrome produced will depend on the degree of physical dependence and the dose of the antagonist administered. Use of a narcotic antagonist in such a person should be avoided. If necessary to treat serious respiratory depression in the physically dependent patient the antagonist should be administered with extreme care and by titration with smaller than usual doses of the antagonist.
Supportive measures (including oxygen, vasopressors) should be employed in the management of circulatory shock and pulmonary edema accompanying overdose as indicated. Cardiac arrest or arrhythmias may require cardiac massage or defibrillation.
(See also: CLINICAL PHARMACOLOGY, AND PRECAUTIONS sections)
Dosage of morphine is a patient-dependent variable, which must be individualized according to patient metabolism, age and disease state and also response to morphine. Each patient should be maintained at the lowest dosage level that will produce acceptable analgesia. As the patient' well-being improves after successful relief of moderate to severe pain, periodic reduction of dosage and/or extension of dosing interval should be attempted to minimize exposure to morphine.
Usual Adult Oral Dose: to 30 mg every four (4) hours or as directed by physician, administered either as MSIR Oral Solutions, MSIR Oral Tablets or MSIR Oral Capsules. For control of pain in terminal illness, it is recommended that the appropriate dose of MSIR Oral Solutions, MSIR Oral Tablets or MSIR Oral Capsules be given on a regularly scheduled basis every four hours at the minimum dose to achieve acceptable analgesia. If converting a patient from another narcotic to morphine sulfate on the basis of standard equivalence tables, a 1 to 3 ratio of parenteral to oral morphine equivalence is suggested. This ratio is conservative and may underestimate the amount of morphine required. If this is the case, the dose of MSIR Oral Solutions, MSIR Oral Tablets or MSIR Oral Capsules should be gradually increased to achieve acceptable analgesia and tolerable side effects.
Sprinkling Contents of Capsule on Food or Liquids: MSIR Oral Capsules may be carefully opened and the entire beaded contents added to a small amount of cool, soft food, such as applesauce or pudding, or a liquid, such as water or orange juice. The bead-food mixture should be swallowed immediately and not stored for future use.
NDC 0034-0521-02: high density polyethylene plastic bottle of 120 mL with child-resistant closure.
NDC 0034-0522-02: high density polyethylene plastic bottle of 120 mL with child-resistant closure.
(unflavored)
NDC 0034-0523-01: high density polyethylene plastic, child-resistant closure bottle with child-resistant dropper in 30 mL size.
NDC 0034-0523-02: high density polyethylene plastic, child-resistant closure bottle with child-resistant dropper in 120 mL size.
Discard opened bottle of Oral Solution after 90 days. Protect from light.
15 mg round, white scored tablets
NDC 0034-0518-10: opaque plastic bottle containing 100 tablets. Each tablet bears the symbol PF on the scored side and MI 15 on the other side.
NDC 0034-0519-10: opaque plastic bottle containing 100 tablets. Each tablet bears the symbol PF on the scored side and MI 30 on the other side.
15 mg capsules, white opaque capsule body with blue cap
NDC 0034-1025-10: opaque plastic bottle containing 100 capsules. Each capsule bears the symbols " PF MSIR 15 " and " THIS END UP. "
NDC 0034-1026-10: opaque plastic bottle containing 100 capsules. Each capsule bears the symbols " PF MSIR 30 " and " THIS END UP. "
Store MSIR Oral Solutions, Tablets and Capsules at controlled room temperature 15°to 30°C (59°- 86°F).
CAUTION: DEA Order Form Required.
Rx Only
THE PURDUE FREDERICK COMPANY
Stamford, CT 06901-3431
Copyright© 1985, 1998
The Purdue Frederick Company
June 9, 2000 I3154
 |
 |
 |