 |
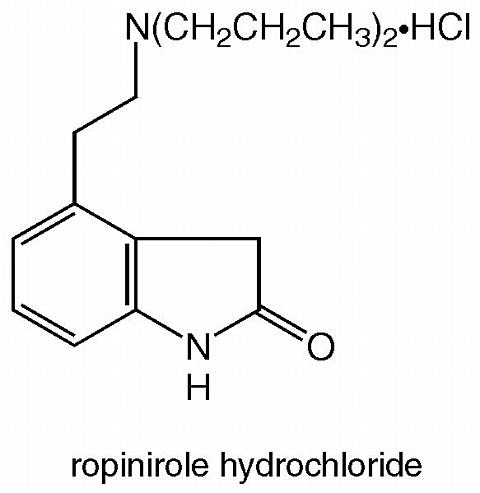
Requip (ropinirole hydrochloride), an orally administered anti-Parkinsonian drug, is a non-ergoline dopamine agonist. It is the hydrochloride salt of 4-[2-(dipropylamino)ethyl]-1,3-dihydro-2H-indol-2-one monohydrochloride and has an empirical formula of C 16 H 24 N 2 O·HCl. The molecular weight is 296.84 (260.38 as the free base).
The structural formula is:
 |
Ropinirole hydrochloride is a white to pale greenish-yellow powder with a melting range of 243° to 250°C and a solubility of 133 mg/mL in water.
Each pentagonal film-coated Tiltab® tablet with beveled edges contains ropinirole hydrochloride equivalent to ropinirole, 0.25 mg, 0.5 mg, 1 mg, 2 mg, 4 mg or 5 mg. Inactive ingredients consist of: croscarmellose sodium, hydrous lactose, magnesium stearate, microcrystalline cellulose, and one or more of the following: FD&C Blue No. 2 aluminum lake, FD&C Yellow No. 6 aluminum lake, hydroxypropyl methylcellulose, iron oxides, polyethylene glycol, polysorbate 80, titanium dioxide.
Requip is a non-ergoline dopamine agonist with high relative in vitro specificity and full intrinsic activity at the D 2 and D 3 dopamine receptor subtypes, binding with higher affinity to D 3 than to D 2 or D 4 receptor subtypes. The relevance of D 3 receptor binding in Parkinson' disease is unknown.
Ropinirole has moderate in vitro affinity for opioid receptors. Ropinirole and its metabolites have negligible in vitro affinity for dopamine D 1 , 5-HT 1 , 5-HT 2 , benzodiazepine, GABA, muscarinic, alpha 1 -, alpha 2 -, and beta-adrenoreceptors.
The precise mechanism of action of Requip as a treatment for Parkinson' disease is unknown, although it is believed to be due to stimulation of post-synaptic dopamine D 2 -type receptors within the caudate-putamen in the brain. This conclusion is supported by studies that show that ropinirole improves motor function in various animal models of Parkinson' disease. In particular, ropinirole attenuates the motor deficits induced by lesioning the ascending nigrostriatal dopaminergic pathway with the neurotoxin 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP) in primates.
In healthy normotensive subjects, single oral doses of Requip in the range 0.01 to 2.5 mg had little or no effect on supine blood pressure and pulse rates. Upon standing, Requip caused decreases in systolic and diastolic blood pressure at doses above 0.25 mg. In some subjects, these changes were associated with the emergence of orthostatic symptoms, bradycardia and, in one case, transient sinus arrest with syncope. The effect of repeat dosing and slow titration of Requip was not studied in healthy volunteers.
The mechanism of Requip -induced postural hypotension is presumed to be due to a D 2 -mediated blunting of the noradrenergic response to standing and subsequent decrease in peripheral vascular resistance. Nausea is a common concomitant of orthostatic signs and symptoms.
At oral doses as low as 0.2 mg, Requip suppressed serum prolactin concentrations in healthy male volunteers.
Requip had no dose-related effect on ECG wave form and rhythm in young healthy male volunteers in the range of 0.01 to 2.5 mg.
Ropinirole is rapidly absorbed after oral administration, reaching peak concentration in approximately 1-2 hours. In clinical studies, over 88% of a radiolabeled dose was recovered in urine and the absolute bioavailability was 55%, indicating a first pass effect. Relative bioavailability from a tablet compared to an oral solution is 85%. Food does not affect the extent of absorption of ropinirole, although its T max is increased by 2.5 hours when the drug is taken with a meal. The clearance of ropinirole after oral administration to patients is 47 L/hr (cv=45%) and its elimination half-life is approximately 6 hours. Ropinirole is extensively metabolized by the liver to inactive metabolites and displays linear kinetics over the therapeutic dosing range of 1 mg to 8 mg t.i.d. Steady-state concentrations are expected to be achieved within 2 days of dosing. Accumulation upon multiple dosing is predictive from single dosing.
Ropinirole is widely distributed throughout the body, with an apparent volume of distribution of 7.5 L/kg (cv=32%). It is up to 40% bound to plasma proteins and has a blood-to-plasma ratio of 1:1.
The major metabolic pathways are N-despropylation and hydroxylation to form the inactive N-despropyl and hydroxy metabolites. In vitro studies indicate that the major cytochrome P 450 isozyme involved in the metabolism of ropinirole is CYP1A2, an enzyme known to be stimulated by smoking and omeprazole, and inhibited by, for example, fluvoxamine, mexiletine, and the older fluoroquinolones, such as ciprofloxacin and norfloxacin. The N-despropyl metabolite is converted to carbamyl glucuronide, carboxylic acid, and N-despropyl hydroxy metabolites. The hydroxy metabolite of ropinirole is rapidly glucuronidated. Less than 10% of the administered dose is excreted as unchanged drug in urine. N-despropyl ropinirole is the predominant metabolite found in urine (40%), followed by the carboxylic acid metabolite (10%), and the glucuronide of the hydroxy metabolite (10%).
P 450 Interaction In vitro metabolism studies showed that CYP1A2 was the major enzyme responsible for the metabolism of ropinirole. There is thus the potential for inhibitors or substrates of this enzyme to alter its clearance when coadministered with ropinirole. Therefore, if therapy with a drug known to be a potent inhibitor of CYP1A2 is stopped or started during treatment with Requip , adjustment of the Requip dose may be required.
Because therapy with Requip is initiated at a subtherapeutic dosage and gradually titrated upward according to clinical tolerability to obtain the optimum therapeutic effect, adjustment of the initial dose based on gender, weight or age is not necessary.
Age: Oral clearance of ropinirole is reduced by 30% in patients above 65 years of age compared to younger patients. Dosage adjustment is not necessary in the elderly (above 65 years) as the dose of ropinirole is to be individually titrated to clinical response.
Gender: Female and male patients showed similar oral clearance.
Race: The influence of race on the pharmacokinetics of ropinirole has not been evaluated.
Cigarette Smoking: The effect of smoking on the oral clearance of ropinirole has not been evaluated. Smoking is expected to increase the clearance of ropinirole since CYP1A2 is known to be induced by smoking.
Renal Impairment: Based on population pharmacokinetic analysis, no difference was observed in the pharmacokinetics of ropinirole in patients with moderate renal impairment (creatinine clearance between 30 to 50 mL/min.) compared to an age-matched population with creatinine clearance above 50 mL/min. Therefore, no dosage adjustment is necessary in moderately renally impaired patients. The use of Requip (ropinirole hydrochloride) in patients with severe renal impairment has not been studied.
The effect of hemodialysis on drug removal is not known, but because of the relatively high apparent volume of distribution of ropinirole (525 L), the removal of the drug by hemodialysis is unlikely.
Hepatic Impairment: The pharmacokinetics of ropinirole have not been studied in hepatically impaired patients. These patients may have higher plasma levels and lower clearance of the drug than patients with normal hepatic function. The drug should be titrated with caution in this population.
Other Diseases: Population pharmacokinetic analysis revealed no change in the oral clearance of ropinirole in patients with concomitant diseases, such as hypertension, depression, osteoporosis/arthritis, and insomnia, compared to patients with Parkinson' disease only.
The effectiveness of Requip in the treatment of Parkinson' disease was evaluated in a multi-national drug development program consisting of 11 randomized, controlled trials. Four were conducted in patients with early Parkinson' disease and no concomitant L-dopa and 7 were conducted in patients with advanced Parkinson' disease with concomitant L-dopa.
Among these 11 studies, three placebo-controlled studies provide the most persuasive evidence of ropinirole's effectiveness in the management of patients with Parkinson' disease who were and were not receiving concomitant L-dopa. Two of these three trials enrolled patients with early Parkinson' disease (without L-dopa) and one enrolled patients receiving L-dopa.
In these studies a variety of measures were used to assess the effects of treatment (e.g., the Unified Parkinson' Disease Rating Scale [UPDRS], Clinical Global Impression scores, patient diaries recording time "on" and "off," and tolerability of L-dopa dose reductions).
In both studies of early Parkinson' disease (without L-dopa) patients, the motor component (Part III) of the UPDRS was the primary outcome assessment. The UPDRS is a four-part multi-item rating scale intended to evaluate mentation (Part I), activities of daily living (Part II), motor performance (Part III), and complications of therapy (Part IV). Part III of the UPDRS contains 14 items designed to assess the severity of the cardinal motor findings in patients with Parkinson' disease (e.g., tremor, rigidity, bradykinesia, postural instability, etc.) scored for different body regions and has a maximum (worst) score of 108. Responders were defined as patients with at least a 30% reduction in the Part III score.
In the study of advanced Parkinson' disease (with L-dopa) patients, both reduction in percent awake time spent "off" and the ability to reduce the daily use of L-dopa were assessed as a combined endpoint and individually.
One early therapy study was a 12-week multicenter study in which 63 patients (41 on Requip ) with idiopathic Parkinson' disease receiving concomitant anti-Parkinson medication (but not L-dopa) were randomized to either Requip or placebo. Patients had a mean disease duration of approximately 2 years. Patients were eligible for enrollment if they presented with bradykinesia and at least tremor, rigidity, or postural instability. In addition, they must have been classified as Hoehn & Yahr Stage I-IV. This scale, ranging from I=unilateral involvement with minimal impairment to V=confined to wheelchair or bed, is a standard instrument used for staging patients with Parkinson' disease. The primary outcome measure in this trial was the proportion of patients experiencing a decrease (compared to baseline) of at least 30% in the UPDRS motor score.
Patients were titrated for up to 10 weeks, starting at 0.5 mg b.i.d., with weekly increments of 0.5 mg b.i.d. to a maximum of 5 mg b.i.d. Once patients reached their maximally tolerated dose (or 5 mg b.i.d.), they were maintained on that dose through 12 weeks. The mean dose achieved by patients at study endpoint was 7.4 mg/day. At the end of 12 weeks, 71% of Requip -treated patients were responders, compared with 41% of patients in the placebo group (p=0.021).
Statistically significant differences between the percentage of responders on Requip compared to placebo were seen after 8 weeks of treatment.
In addition, the mean percentage improvement from baseline in the Total Motor Score was 43% in Requip -treated patients compared with 21% in placebo-treated patients (p=0.018).
Statistically significant differences in UPDRS motor score between Requip and placebo were seen after 2 weeks of treatment.
The median daily dose at which a 30% reduction in UPDRS motor score was sustained was 4 mg.
The second trial in early Parkinson' disease (without L-dopa) patients was a double-blind, randomized, placebo-controlled 6-month study. Patients were essentially similar to those in the study described above; concomitant use of selegiline was allowed, but patients were not permitted to use anticholinergics or amantadine during the study. Patients had a mean disease duration of 2 years and limited (not more than a 6-week period) or no prior exposure to L-dopa. The starting dose of Requip in this trial was 0.25 mg t.i.d. The dose was titrated at weekly intervals by increments of 0.25 mg t.i.d. to a dose of 1.0 mg t.i.d. Further titrations at weekly intervals were at increments of 0.5 mg t.i.d. up to a dose of 3.0 mg t.i.d and then weekly at increments of 1.0 mg t.i.d. Patients were to be titrated to a dose of at least 1.5 mg t.i.d. and then to their maximally tolerated dose, up to a maximum of 8.0 mg t.i.d. The mean dose attained in patients at study endpoint was 15.7 mg/day.
The primary measure of effectiveness was the mean percent reduction (improvement) from baseline in the UPDRS Motor Score. In this study 241 patients were enrolled. At the end of the 6-month study, Requip -treated patients had 22% improvement in motor score, compared with a 4% worsening in the placebo group (p<0.001).
Statistically significant differences in UPDRS motor score improvement between Requip and placebo were seen after 12 weeks of treatment.
This double-blind, randomized, placebo-controlled 6-month trial evaluated 148 patients (Hoehn & Yahr II-IV) who were not adequately controlled on L-dopa. Patients in this study had a mean disease duration of approximately 9 years, had been exposed to L-dopa for approximately 7 years, and had experienced "on-off" periods with L-dopa therapy. Patients previously receiving stable doses of selegiline, amantadine and/or anticholinergic agents could continue on these agents during the study. Patients were started at a Requip dose of 0.25 mg t.i.d. and titrated upward by weekly intervals until an optimal therapeutic response was achieved. The maximum dose of study medication was 8 mg t.i.d. All patients had to be titrated to at least a dose of 2.5 mg t.i.d. Patients could then be maintained on this dose level or higher for the remainder of the study. Once a dose of 2.5 mg t.i.d. was achieved, patients underwent a mandatory reduction in their L-dopa dose, to be followed by additional mandatory reductions with continued escalation of the Requip dose. Reductions in the dosage of L-dopa were also allowed if patients experienced adverse events that the investigator considered related to dopaminergic therapy. The mean dose attained at study endpoint was 16.3 mg/day. The primary outcome was the proportion of responders, defined as patients who were able both to achieve a decrease (compared to baseline) of at least 20% in their L-dopa dose and a decrease of at least 20% in the proportion of the time awake in the "off" condition (a period of time during the day when patients are particularly immobile), as determined by patient diary. In addition, the mean percent change from baseline in daily L-dopa dose was examined.
At the end of 6 months, 28% of Requip -treated patients were classified as responders (based on combined endpoint) while 11% of placebo-treated patients were responders (p=0.02). Based on the protocol-mandated reductions in L-dopa dosage with escalating Requip doses, Requip -treated patients had a 19.4% mean reduction in L-dopa dose while placebo-treated patients had a 3% reduction (p<0.001). L-dopa dosage reduction was also allowed during the study if dyskinesias or other dopaminergic effects occurred. Overall, reduction of L-dopa dose was sustained in 87% of Requip -treated patients and in 57% of patients on placebo. On average, the L-dopa dose was reduced by 31% in Requip -treated patients.
The mean number of "off" hours per day during baseline was 6.4 hours for Requip -treated patients and 7.3 hours for patients treated with placebo. At the end of the 6-month study, patients treated with Requip had a mean of 4.9 hours per day of "off" time, while placebo-treated patients had a mean of 6.4 hours per day of "off" time.
Requip (ropinirole hydrochloride) is indicated for the treatment of the signs and symptoms of idiopathic Parkinson' disease.
The effectiveness of Requip was demonstrated in randomized, controlled trials in patients with early Parkinson's disease who were not receiving concomitant L-dopa therapy as well as in patients with advanced disease on concomitant L-dopa (see , Clinical Trials ).
Requip is contraindicated for patients known to have hypersensitivity to the product.
Patients treated with Requip have reported falling asleep while engaged in activities of daily living, including the operation of motor vehicles which sometimes resulted in accidents. Although many of these patients reported somnolence while on Requip , some perceived that they had no warning signs such as excessive drowsiness, and believed that they were alert prior to the event. Some of these events have been reported as late as one year after initiation of treatment.
Somnolence is a common occurrence in patients receiving Requip . Many clinical experts believe that falling asleep while engaged in activities of daily living always occurs in a setting of pre-existing somnolence although patients may not give such a history. For this reason, prescribers should continually reassess patients for drowsiness or sleepiness especially since some of the events occur well after the start of treatment. Prescribers should also be aware that patients may not acknowledge drowsiness or sleepiness until directly questioned about drowsiness or sleepiness during specific activities.
Before initiating treatment with Requip , patients should be advised of the potential to develop drowsiness and specifically asked about factors that may increase the risk with Requip such as concomitant sedating medications, the presence of sleep disorders, and concomitant medications that increase ropinirole plasma levels (e.g., ciprofloxacin--see PRECAUTIONS , Drug Interactions ). If a patient develops significant daytime sleepiness or episodes of falling asleep during activities that require active participation (e.g., conversations, eating, etc.), Requip should ordinarily be discontinued. [See DOSAGE AND ADMINISTRATION for guidance in discontinuing Requip .] If a decision is made to continue Requip , patients should be advised to not drive and to avoid other potentially dangerous activities. There is insufficient information to establish that dose reduction will eliminate episodes of falling asleep while engaged in activities of daily living.
Syncope, sometimes associated with bradycardia, was observed in association with ropinirole in both early Parkinson' disease (without L-dopa) patients and advanced Parkinson' disease (with L-dopa) patients. In the two double-blind placebo-controlled studies of Requip in patients with Parkinson' disease who were not being treated with L-dopa, 11.5% (18 of 157) of patients on Requip had syncope compared to 1.4% (2 of 147) of patients on placebo. Most of these cases occurred more than 4 weeks after initiation of therapy with Requip , and were usually associated with a recent increase in dose.
Of 208 patients being treated with both L-dopa and Requip , in placebo-controlled advanced Parkinson' disease trials, there were reports of syncope in 6 (2.9%) compared to 2 of 120 (1.7%) of placebo/L-dopa patients.
Because the studies of Requip excluded patients with significant cardiovascular disease, it is not known to what extent the estimated incidence figures apply to Parkinson' disease patients as a whole. Therefore, patients with severe cardiovascular disease should be treated with caution.
Two of 47 Parkinson' disease patient volunteers enrolled in phase 1 studies had syncope following a 1 mg dose. In phase 1 studies including 110 healthy volunteers, one patient developed hypotension, bradycardia, and sinus arrest of 26 seconds accompanied by syncope; the patient recovered spontaneously without intervention. One other healthy volunteer reported syncope.
Dopamine agonists, in clinical studies and clinical experience, appear to impair the systemic regulation of blood pressure, with resulting postural hypotension, especially during dose escalation. Parkinson's disease patients, in addition, appear to have an impaired capacity to respond to a postural challenge. For these reasons, Parkinson's patients being treated with dopaminergic agonists ordinarily (1) require careful monitoring for signs and symptoms of postural hypotension, especially during dose escalation, and (2) should be informed of this risk (see PRECAUTIONS , Information for Patients ).
Although the clinical trials were not designed to systematically monitor blood pressure, there were individual reported cases of postural hypotension in early Parkinson' disease (without L-dopa) Requip -treated patients. Most of these cases occurred more than 4 weeks after initiation of therapy with Requip , and were usually associated with a recent increase in dose.
In phase 1 studies of Requip that included 110 healthy volunteers, nine subjects had documented symptomatic postural hypotension. These episodes appeared mainly at doses above 0.8 mg and these doses are higher than the starting doses recommended for Parkinson' disease patients. In eight of these nine individuals, the hypotension was accompanied by bradycardia, but did not develop into syncope. (See Syncope above.) None of these events resulted in death or hospitalization.
One of 47 Parkinson' disease patient volunteers enrolled in phase 1 studies had documented hypotension following a 2 mg dose on two occasions.
In double-blind, placebo-controlled, early therapy studies in patients with Parkinson' disease who were not treated with L-dopa, 5.2% (8 of 157) of patients treated with Requip reported hallucinations, compared to 1.4% of patients on placebo (2 of 147). Among those patients receiving both Requip and L-dopa, in advanced Parkinson' disease (with L-dopa) studies, 10.1% (21 of 208) were reported to experience hallucinations, compared to 4.2% (5 of 120) of patients treated with placebo and L-dopa.
Hallucinations were of sufficient severity to cause discontinuation of treatment in 1.3% of the early Parkinson' disease (without L-dopa) patients and 1.9% of the advanced Parkinson' disease (with L-dopa) patients compared to 0% and 1.7% of placebo patients, respectively.
General
Dyskinesia: Requip may potentiate the dopaminergic side effects of L-dopa and may cause and/or exacerbate pre-existing dyskinesia. Decreasing the dose of L-dopa may ameliorate this side effect.
Renal and Hepatic: No dosage adjustment is needed in patients with mild to moderate renal impairment (creatinine clearance of 30 to 50 mL/min.). Because the use of Requip in patients with severe renal or hepatic impairment has not been studied, administration of Requip to such patients should be carried out with caution.
Withdrawal Emergent Hyperpyrexia and Confusion: Although not reported with Requip , a symptom complex resembling the neuroleptic malignant syndrome (characterized by elevated temperature, muscular rigidity, altered consciousness, and autonomic instability), with no other obvious etiology, has been reported in association with rapid dose reduction, withdrawal of, or changes in anti-Parkinsonian therapy.
Fibrotic Complications: Cases of retroperitoneal fibrosis, pulmonary infiltrates, pleural effusion, and pleural thickening have been reported in some patients treated with ergot-derived dopaminergic agents. While these complications may resolve when the drug is discontinued, complete resolution does not always occur.
Although these adverse events are believed to be related to the ergoline structure of these compounds, whether other, nonergot derived dopamine agonists can cause them is unknown.
In the Requip development program, a 69-year-old man with obstructive lung disease was treated with Requip for 16 months and developed pleural thickening and effusion accompanied by lower extremity edema, cardiomegaly, pleuritic pain, and shortness of breath. Pleural biopsy demonstrated chronic inflammation and sclerosis. The effusion resolved after medical therapy and discontinuation of Requip . The patient was lost to follow-up. The relationship of these events to Requip (ropinirole hydrochloride) cannot be established.
Retinal pathology in albino rats: Retinal degeneration was observed in albino rats in the 2-year carcinogenicity study at all doses tested (equivalent to 0.6 to 20 times the maximum recommended human dose on a mg/m 2 basis), but was statistically significant at the highest dose (50 mg/kg/day). Additional studies to further evaluate the specific pathology (e.g., loss of photoreceptor cells) have not been performed. Similar changes were not observed in a 2-year carcinogenicity study in albino mice or in rats or monkeys treated for 1 year.
The potential significance of this effect in humans has not been established, but cannot be disregarded because disruption of a mechanism that is universally present in vertebrates (e.g., disk shedding) may be involved.
Binding to melanin: Requip binds to melanin-containing tissues (i.e., eyes, skin) in pigmented rats. After a single dose, long-term retention of drug was demonstrated, with a half-life in the eye of 20 days. It is not known if Requip accumulates in these tissues over time.
Patients should be instructed to take Requip only as prescribed.
Requip can be taken with or without food. Since ingestion with food reduces the maximum concentration (C max ) of Requip , patients should be advised that taking Requip with food may reduce the occurrence of nausea. However, this has not been established in controlled clinical trials.
Patients should be informed that hallucinations can occur, and that the elderly are at a higher risk than younger patients with Parkinson' disease.
Patients should be advised that they may develop postural (orthostatic) hypotension with or without symptoms such as dizziness, nausea, syncope, and sometimes sweating. Hypotension and/or orthostatic symptoms may occur more frequently during initial therapy or with an increase in dose at any time (cases have been seen after weeks of treatment). Accordingly, patients should be cautioned against rising rapidly after sitting or lying down, especially if they have been doing so for prolonged periods, and especially at the initiation of treatment with Requip .
Patients should be alerted to the potential sedating effects associated with Requip including somnolence and the possibility of falling asleep while engaged in activities of daily living. Since somnolence is a frequent adverse event with potentially serious consequences, patients should neither drive a car nor engage in other potentially dangerous activities until they have gained sufficient experience with Requip to gauge whether or not it affects their mental and/or motor performance adversely. Patients should be advised that if increased somnolence or episodes of falling asleep during activities of daily living (e.g., watching television, passenger in a car, etc.) are experienced at any time during treatment, they should not drive or participate in potentially dangerous activities until they have contacted their physician. Because of possible additive effects, caution should be advised when patients are taking other sedating medications or alcohol in combination with Requip and when taking concomitant medications that increase plasma levels of ropinirole (e.g., ciprofloxacin).
Because of the possible additive sedative effects, caution should also be used when patients are taking alcohol or other CNS depressants (e.g., benzodiazepines, antipsychotics, antidepressants, etc.) in combination with Requip .
Because of the possibility that ropinirole may be excreted in breast milk, patients should be advised to notify their physicians if they intend to breast-feed or are breast-feeding an infant.
Because ropinirole has been shown to have adverse effects on embryo-fetal development, including teratogenic effects, in animals, and because experience in humans is limited, patients should be advised to notify their physician if they become pregnant or intend to become pregnant during therapy (see PRECAUTIONS , Pregnancy).
P 450 Interaction: In vitro metabolism studies showed that CYP1A2 was the major enzyme responsible for the metabolism of ropinirole. There is thus the potential for substrates or inhibitors of this enzyme when coadministered with ropinirole to alter its clearance. Therefore, if therapy with a drug known to be a potent inhibitor of CYP1A2 is stopped or started during treatment with Requip , adjustment of the Requip dose may be required.
L-dopa: Co-administration of carbidopa + L-dopa (Sinemet® 10/100 mg b.i.d.) with ropinirole (2.0 mg t.i.d.) had no effect on the steady-state pharmacokinetics of ropinirole (n=28 patients). Oral administration of Requip 2.0 mg t.i.d. increased mean steady state C max of L-dopa by 20% but its AUC was unaffected (n=23 patients).
Digoxin Co-administration of Requip (2.0 mg t.i.d.) with digoxin (0.125-0.25 mg q.d.) did not alter the steady-state pharmacokinetics of digoxin in 10 patients.
Theophylline Administration of theophylline (300 mg b.i.d., a substrate of CYP1A2) did not alter the steady-state pharmacokinetics of ropinirole (2 mg t.i.d.) in 12 patients with Parkinson' disease. Ropinirole (2 mg t.i.d.) did not alter the pharmacokinetics of theophylline (5 mg/kg i.v.) in 12 patients with Parkinson' disease.
Ciprofloxacin: Co-administration of ciprofloxacin (500 mg b.i.d.), an inhibitor of CYP1A2, with ropinirole (2 mg t.i.d.) increased ropinirole AUC by 84% on average, and C max by 60% (n=12 patients).
Estrogens: Population pharmacokinetic analysis revealed that estrogens (mainly ethinylestradiol: intake 0.6-3 mg over 4-month to 23-year period) reduced the oral clearance of ropinirole by 36% in 16 patients. Dosage adjustment may not be needed for Requip in patients on estrogen therapy because patients must be carefully titrated with ropinirole to tolerance or adequate effect. However, if estrogen therapy is stopped or started during treatment with Requip , then adjustment of the Requip (ropinirole hydrochloride) dose may be required.
Dopamine Antagonists: Since ropinirole is a dopamine agonist, it is possible that dopamine antagonists, such as neuroleptics (phenothiazines, butyrophenones, thioxanthenes) or metoclopramide, may diminish the effectiveness of Requip . Patients with major psychotic disorders, treated with neuroleptics, should only be treated with dopamine agonists if the potential benefits outweigh the risks.
Population analysis showed that commonly administered drugs, e.g., selegiline, amantadine, tricyclic antidepressants, benzodiazepines, ibuprofen, thiazides, antihistamines, and anticholinergics did not affect the oral clearance of ropinirole.
Two-year carcinogenicity studies were conducted in Charles River CD-1 mice at doses of 5, 15, and 50 mg/kg/day and in Sprague-Dawley rats at doses of 1.5, 15, and 50 mg/kg/day (top doses equivalent to 10 times and 20 times, respectively, the maximum recommended human dose of 24 mg/day on a mg/m 2 basis). In the male rat, there was a significant increase in testicular Leydig cell adenomas at all doses tested, i.e., >/=1.5 mg/kg (0.6 times the maximum recommended human dose on a mg/m 2 basis). This finding is of questionable significance because the endocrine mechanisms believed to be involved in the production of Leydig cell hyperplasia and adenomas in rats are not relevant to humans. In the female mouse, there was an increase in benign uterine endometrial polyps at a dose of 50 mg/kg/day (10 times the maximum recommended human dose on a mg/m 2 basis
Ropinirole was not mutagenic or clastogenic in the in vitro Ames test, the in vitro chromosome aberration test in human lymphocytes, the in vitro mouse lymphoma (L1578Y cells) assay, and the in vivo mouse micronucleus test.
When administered to female rats prior to and during mating and throughout pregnancy, ropinirole caused disruption of implantation at doses of 20 mg/kg/day (8 times the maximum recommended human dose on a mg/m 2 basis) or greater. This effect is thought to be due to the prolactin-lowering effect of ropinirole. In humans, chorionic gonadotropin, not prolactin, is essential for implantation. In rat studies using low doses (5 mg/kg) during the prolactin-dependent phase of early pregnancy (gestation days 0-8), ropinirole did not affect female fertility at dosages up to 100 mg/kg/day (40 times the maximum recommended human dose on a mg/m 2 basis). No effect on male fertility was observed in rats at dosages up to 125 mg/kg/day (50 times the maximum recommended human dose on a mg/m 2 basis
Pregnancy Category C: In animal reproduction studies, ropinirole has been shown to have adverse effects on embryo-fetal development, including teratogenic effects. Ropinirole given to pregnant rats during organogenesis (20 mg/kg on gestation days 6 and 7 followed by 20, 60, 90, 120 or 150 mg/kg on gestation days 8 through 15) resulted in decreased fetal body weight at 60 mg/kg/day, increased fetal death at 90 mg/kg/day, and digital malformations at 150 mg/kg/day (24, 36 and 60 times the maximum recommended clinical dose on a mg/m 2 basis, respectively). The combined administration of ropinirole (10 mg/kg/day; 8 times the maximum recommended human dose on a mg/m 2 basis) and L-dopa (250 mg/kg/day) to pregnant rabbits during organogenesis produced a greater incidence and severity of fetal malformations (primarily digit defects) than were seen in the offspring of rabbits treated with L-dopa alone. No indication of an effect on development of the conceptus was observed in rabbits when a maternally toxic dose of ropinirole was administered alone (20 mg/kg/day: 16 times the maximum recommended human dose on a mg/m 2 basis). In a perinatal-postnatal study in rats, 10 mg/kg/day (4 times the maximum recommended human dose on a mg/m 2 basis) of ropinirole impaired growth and development of nursing offspring and altered neurological development of female offspring.
There are no adequate and well-controlled studies using Requip in pregnant women. Requip should be used during pregnancy only if the potential benefit outweighs the potential risk to the fetus.
Requip inhibits prolactin secretion in humans and could potentially inhibit lactation.
Studies in rats have shown that Requip and/or its metabolite(s) is excreted in breast milk. It is not known whether this drug is excreted in human milk. Because many drugs are excreted in human milk and because of the potential for serious adverse reactions in nursing infants from Requip , a decision should be made whether to discontinue nursing or to discontinue the drug, taking into account the importance of the drug to the mother.
Safety and effectiveness in the pediatric population have not been established.
During the pre-marketing development of Requip , patients received Requip either without L-dopa (early Parkinson' disease studies) or as concomitant therapy with L-dopa (advanced Parkinson' disease studies). Because these 2 populations may have differential risks for various adverse events, this section will, in general, present adverse event data for these 2 populations separately.
The most commonly observed adverse events (>5%) in the double-blind, placebo-controlled early Parkinson' disease trials associated with the use of Requip (n=157) not seen at an equivalent frequency among the placebo-treated patients (n=147) were, in order of decreasing incidence: nausea, dizziness, somnolence, headache, vomiting, syncope, fatigue, dyspepsia, viral infection, constipation, pain, increased sweating, asthenia, dependent/leg edema, orthostatic symptoms, abdominal pain, pharyngitis, confusion, hallucinations, urinary tract infections, and abnormal vision.
Approximately 24% of 157 Requip -treated patients who participated in the double-blind, placebo-controlled early Parkinson' disease (without L-dopa) trials discontinued treatment due to adverse events compared to 13% of 147 patients who received placebo. The adverse events most commonly causing discontinuation of treatment by Requip -treated patients were: nausea (6.4%), dizziness (3.8%), aggravated Parkinson's disease (1.3%), hallucinations (1.3%), somnolence (1.3%), vomiting (1.3%) and headache (1.3%). Of these, hallucinations appear to be dose-related. While other adverse events leading to discontinuation may be dose-related, the titration design utilized in these trials precluded an adequate assessment of the dose response. For example, in the larger of the 2 trials described in , Clinical Trials , the difference in the rate of discontinuations emerged only after 10 weeks of treatment, suggesting, although not proving, that the effect could be related to dose.
Table 1 lists treatment-emergent adverse events that occurred in >/=2% of patients with early Parkinson' disease (without L-dopa) treated with Requip participating in the double-blind, placebo-controlled studies and were numerically more common in the Requip group. In these studies, either Requip (ropinirole hydrochloride) or placebo was used as early therapy (i.e., without L-dopa).
The prescriber should be aware that these figures cannot be used to predict the incidence of adverse events in the course of usual medical practice where patient characteristics and other factors differ from those that prevailed in the clinical studies. Similarly, the cited frequencies cannot be compared with figures obtained from other clinical investigations involving different treatments, uses and investigators. However, the cited figures do provide the prescribing physician with some basis for estimating the relative contribution of drug and non-drug factors to the adverse-events incidence rate in the population studied.
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Other events reported by 1% or more of early Parkinson' disease (without L-dopa) patients treated with Requip , but that were equally or more frequent in the placebo group were: headache, upper respiratory infection, insomnia, arthralgia, tremor, back pain, anxiety, dyskinesias, aggravated Parkinsonism, depression, falls, myalgia, leg cramps, paresthesias, nervousness, diarrhea, arthritis, hot flushes, weight loss, rash, cough, hyperglycemia, muscle spasm, arthrosis, abnormal dreams, dystonia, increased salivation, bradycardia, gout, basal cell carcinoma, gingivitis, hematuria, and rigors.
Among the treatment-emergent adverse events in patients treated with Requip , hallucinations appear to be dose-related.
The incidence of adverse events was not materially different between women and men.
The most commonly observed adverse events (>5%), in the double-blind, placebo-controlled advanced Parkinson' disease (with L-dopa) trials associated with the use of Requip (n = 208) as an adjunct to L-dopa not seen at an equivalent frequency among the placebo-treated patients (n = 120) were, in order of decreasing incidence: dyskinesias, nausea, dizziness, aggravated Parkinsonism, somnolence, headache, insomnia, injury, hallucinations, falls, abdominal pain, upper respiratory infection, confusion, increased sweating, vomiting, viral infection, increased drug level, arthralgia, tremor, anxiety, urinary tract infection, constipation, dry mouth, pain, hypokinesia, and paresthesia.
Approximately 24% of 208 patients who received Requip (ropinirole hydrochloride) in the double-blind, placebo-controlled advanced Parkinson' disease (with L-dopa) trials discontinued treatment due to adverse events compared to 18% of 120 patients who received placebo. The events most commonly (>/=1%) causing discontinuation of treatment by Requip -treated patients were: dizziness (2.9%), dyskinesias (2.4%), vomiting (2.4%), confusion (2.4%), nausea (1.9%), hallucinations (1.9%), anxiety (1.9%), and increased sweating (1.4%). Of these, hallucinations and dyskinesias appear to be dose-related.
Table 2 lists treatment-emergent adverse events that occurred in >/=2% of patients with advanced Parkinson' disease (with L-dopa) treated with Requip who participated in the double-blind, placebo-controlled studies and were numerically more common in the Requip group. In these studies, either Requip or placebo was used as an adjunct to L-dopa. Adverse events were usually mild or moderate in intensity.
The prescriber should be aware that these figures cannot be used to predict the incidence of adverse events in the course of usual medical practice where patient characteristics and other factors differ from those that prevailed in the clinical studies. Similarly, the cited frequencies cannot be compared with figures obtained from other clinical investigations involving different treatments, uses, and investigators. However, the cited figures do provide the prescribing physician with some basis for estimating the relative contribution of drug and non-drug factors to the adverse-events incidence rate in the population studied.
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Other events reported by 1% or more of patients treated with both Requip and L-dopa, but equally or more frequent in the placebo/L-dopa group were: myocardial infarction, orthostatic symptoms, virus infections, asthenia, dyspepsia, myalgia, back pain, depression, leg cramps, fatigue, rhinitis, chest pain, hematuria, vertigo, tinnitus, leg edema, hot flushes, abnormal gait, hyperkinesia, and pharyngitis.
Among the treatment-emergent adverse events in patients treated with Requip , hallucinations and dyskinesias appear to be dose-related.
Other Adverse Events Observed During All Phase 2/3 Clinical Trials: Requip has been administered to 1,599 individuals in clinical trials. During these trials, all adverse events were recorded by the clinical investigators using terminology of their own choosing. To provide a meaningful estimate of the proportion of individuals having adverse events, similar types of events were grouped into a smaller number of standardized categories using modified WHOART dictionary terminology. These categories are used in the listing below. The frequencies presented represent the proportion of the 1,599 individuals exposed to Requip who experienced events of the type cited on at least one occasion while receiving Requip . All reported events that occurred at least twice (or once for serious or potentially serious events), except those already listed above, trivial events, and terms too vague to be meaningful are included, without regard to determination of a causal relationship to Requip (ropinirole hydrochloride), except that events very unlikely to be drug-related have been deleted.
Events are further classified within body system categories and enumerated in order of decreasing frequency using the following definitions: frequent adverse events are defined as those occurring in at least 1/100 patients and infrequent adverse events are those occurring in 1/100 to 1/1000 patients and rare events are those occurring in fewer than 1/1000 patients.
Body as a Whole: infrequent -- cellulitis, peripheral edema, fever, influenza-like symptoms, enlarged abdomen, precordial chest pain, and generalized edema; rare -- ascites.
Cardiovascular: infrequent -- cardiac failure, bradycardia, tachycardia, supraventricular tachycardia, angina pectoris, bundle branch block, cardiac arrest, cardiomegaly, aneurysm, mitral insufficiency; rare -- ventricular tachycardia.
Central/Peripheral Nervous System: frequent -- neuralgia; infrequent -- involuntary muscle contractions, hypertonia, dysphonia, abnormal coordination, extrapyramidal disorder, migraine, choreoathetosis, coma, stupor, aphasia, convulsions, hypotonia, peripheral neuropathy, paralysis; rare -- grand mal convulsions, hemiparesis, hemiplegia.
Endocrine: infrequent -- hypothyroidism, gynecomastia, hyperthyroidism; rare -- goiter, SIADH.
Gastrointestinal: infrequent -- increased hepatic enzymes, bilirubinemia, cholecystitis, cholelithiasis colitis, dysphagia, periodontitis, fecal incontinence, gastroesophageal reflux, hemorrhoids, toothache, eructation, gastritis, esophagitis, hiccups, diverticulitis, duodenal ulcer, gastric ulcer, melena, duodenitis, gastrointestinal hemorrhage, glossitis, rectal hemorrhage, pancreatitis, stomatitis and ulcerative stomatitis, tongue edema; rare -- biliary pain, hemorrhagic gastritis, hematemesis, salivary duct obstruction.
Hematologic: infrequent -- purpura, thrombocytopenia, hematoma, Vitamin B12 deficiency, hypochromic anemia, eosinophilia, leukocytosis, leukopenia, lymphocytosis, lymphopenia, lymphedema.
Metabolic/Nutritional: frequent -- increased BUN; infrequent -- hypoglycemia, increased alkaline phosphatase, increased LDH, weight increase, hyperphosphatemia, hyperuricemia, diabetes mellitus, glycosuria, hypokalemia, hypercholesterolemia, hyperkalemia, acidosis, hyponatremia, thirst, increased CPK, dehydration; rare -- hypochloremia.
Musculoskeletal: infrequent -- aggravated arthritis, tendinitis, osteoporosis, bursitis, polymyalgia rheumatica, muscle weakness, skeletal pain, torticollis; rare -- Dupuytren' contracture requiring surgery.
Neoplasm: infrequent -- malignant breast neoplasm; rare -- bladder carcinoma, benign brain neoplasm, esophageal carcinoma, malignant laryngeal neoplasm, lipoma, rectal carcinoma, uterine neoplasm.
Psychiatric: infrequent -- increased libido, agitation, apathy, impaired concentration, depersonalization, paranoid reaction, personality disorder, euphoria, delirium, dementia, delusion, emotional lability, decreased libido, manic reaction, somnambulism, aggressive reaction, neurosis; rare -- suicide attempt.
Genitourinary infrequent -- amenorrhea, vaginal hemorrhage, penile disorder, prostatic disorder, balanoposthitis, epididymitis, perineal pain, dysuria, micturition frequency, albuminuria, nocturia, polyuria, renal calculus; rare -- breast enlargement, mastitis, uterine hemorrhage, ejaculation disorder, Peyronie' Disease, pyelonephritis, acute renal failure, uremia.
Resistance Mechanism: infrequent -- herpes zoster, otitis media, sepsis, abscess, herpes simplex, fungal infection, genital moniliasis.
Respiratory: infrequent -- asthma, epistaxis, laryngitis, pleurisy, pulmonary edema.
Skin/Appendage: infrequent -- pruritis, dermatitis, eczema, skin ulceration, alopecia, skin hypertrophy, skin discoloration, urticaria, fungal dermatitis, furunculosis, hyperkeratosis, photosensitivity reaction, psoriasis, maculopapular rash, psoriaform rash, seborrhea.
Special Senses: infrequent -- tinnitus, earache, decreased hearing, abnormal lacrimation, conjunctivitis, blepharitis, glaucoma, abnormal accommodation, blepharospasm, eye pain, photophobia; rare -- scotoma.
Vascular Extracardiac: infrequent -- varicose veins, phlebitis, peripheral gangrene; rare -- limb embolism, pulmonary embolism, gangrene, subarachnoid hemorrhage, deep thrombophlebitis, leg thrombophlebitis, thrombosis.
Falling Asleep During Activities of Daily Living: Patients treated with Requip have reported falling asleep while engaged in activities of daily living, including operation of a motor vehicle which sometimes resulted in accidents (see bolded WARNING ).
Requip is not a controlled substance.
Animal studies and human clinical trials with Requip (ropinirole hydrochloride) did not reveal any potential for drug-seeking behavior or physical dependence.
There were no reports of intentional overdose of Requip in the premarketing clinical trials. A total of 27 patients accidentally took more than their prescribed dose of Requip , with 10 patients ingesting more than 24 mg/day. The largest overdose reported in premarketing clinical trials was 435 mg taken over a 7-day period (62.1 mg/day). Of patients who received a dose greater than 24 mg/day, one experienced mild oro-facial dyskinesia, another patient experienced intermittent nausea. Other symptoms reported with accidental overdoses were: agitation, increased dyskinesia, grogginess, sedation, orthostatic hypotension, chest pain, confusion, vomiting and nausea.
It is anticipated that the symptoms of Requip overdose will be related to its dopaminergic activity. General supportive measures are recommended. Vital signs should be maintained, if necessary. Removal of any unabsorbed material (e.g., by gastric lavage) should be considered.
In all clinical studies, dosage was initiated at a subtherapeutic level and gradually titrated to therapeutic response. The dosage should be increased to achieve a maximum therapeutic effect, balanced against the principal side effects of nausea, dizziness, somnolence and dyskinesia.
Requip should be taken three times daily. Requip can be taken with or without food. Since ingestion with food reduces the maximum concentration (C max ) of Requip , patients should be advised that taking Requip with food may reduce the occurrence of nausea. However, this has not been established in controlled clinical trials.
The recommended starting dose is 0.25 mg three times daily. Based on individual patient response, dosage should then be titrated with weekly increments as described in the table below. After week 4, if necessary, daily dosage may be increased by 1.5 mg per day on a weekly basis up to a dose of 9 mg per day, and then by up to 3 mg per day weekly to a total dose of 24 mg per day.
|
Doses greater than 24 mg/day have not been tested in clinical trials.
When Requip is administered as adjunct therapy to L-dopa, the concurrent dose of L-dopa may be decreased gradually as tolerated. L-dopa dosage reduction was allowed during the advanced Parkinson' disease (with L-dopa) study if dyskinesias or other dopaminergic effects occurred. Overall, reduction of L-dopa dose was sustained in 87% of Requip -treated patients and in 57% of patients on placebo. On average the L-dopa dose was reduced by 31% in Requip -treated patients.
Requip should be discontinued gradually over a 7-day period. The frequency of administration should be reduced from three times daily to twice daily for 4 days. For the remaining 3 days, the frequency should be reduced to once daily prior to complete withdrawal of Requip (ropinirole hydrochloride).
Tablets: Each pentagonal film-coated Tiltab® tablet with beveled edges contains ropinirole hydrochloride as follows: 0.25 mg-white imprinted with SB and 4890; 0.5 mg-yellow imprinted with SB and 4891; 1.0 mg-green imprinted with SB and 4892; 2.0 mg-pale yellowish pink imprinted with SB and 4893; 4.0 mg-pale brown imprinted with SB and 4896; 5.0 mg-blue imprinted with SB and 4894.
0.25 mg SUP 30's: NDC 0007-4890-14
0.25 mg bottles of 100: NDC 0007-4890-20
0.5 mg SUP 30's: NDC 0007-4891-14
0.5 mg bottles of 100: NDC 0007-4891-20
1 mg SUP 30's: NDC 0007-4892-14
1 mg bottles of 100: NDC 0007-4892-20
2 mg SUP 30's: NDC 0007-4893-14
2 mg bottles of 100: NDC 0007-4893-20
4 mg bottles of 100: NDC 0007-4896-20
5 mg SUP 30's: NDC 0007-4894-14
5 mg bottles of 100: NDC 0007-4894-20
Protect from light and moisture. Close container tightly after each use.
Store at controlled room temperature 20°-25°C (68°-77°F) [see USP].
Manufactured in Crawley, UK by SmithKline Beecham Pharmaceuticals , for
SmithKline Beecham Pharmaceuticals , Philadelphia, PA 19101
RQ:L8 Rx only
 |