 |
TERAZOL® 3 (terconazole) Vaginal Cream 0.8% is a white to off-white, water washable cream for intravaginal administration containing 0.8% of the antifungal agent terconazole, cis -1-[ p -[[2-(2,4-Dichlorophenyl)-2-(1H-1,2,4-triazol-1-ylmethyl)-1,3-dioxolan-4-yl] methoxy] phenyl]-4-isopropylpiperazine, compounded in a cream base consisting of butylated hydroxyanisole, cetyl alcohol, isopropyl myristate, polysorbate 60, polysorbate 80, propylene glycol, stearyl alcohol, and purified water.
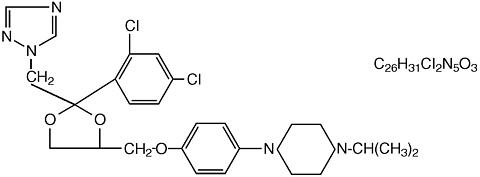
The structural formula of terconazole is as follows:
 |
Terconazole, a triazole derivative, is a white to almost white powder with a molecular weight of 532.47. It is insoluble in water; sparingly soluble in ethanol; and soluble in butanol.
Following daily intravaginal administration of 0.8% terconazole 40 mg (0.8% cream × 5 g) for seven days to normal humans, plasma concentrations were low and gradually rose to a daily peak (mean of 5.9 ng/mL or 0.006 mcg/mL) at 6.6 hours. Results from similar studies in patients with vulvovaginal candidiasis indicate that the slow rate of absorption, the lack of accumulation, and the mean peak plasma concentration of terconazole was not different from that observed in healthy women. The absorption characteristics of terconazole 0.8% in pregnant or nonpregnant patients with vulvovaginal candidiasis were also similar to those found in normal volunteers.
Following oral (30 mg) administration of 14 C-labelled terconazole, the harmonic half-life of elimination from the blood for the parent terconazole was 6.9 hours (range 4.0-11.3). Terconazole is extensively metabolized; the plasma AUC for terconazole compared to the AUC for total radioactivity was 0.6%. Total radioactivity was eliminated from the blood with a harmonic half-life of 52.2 hours (range 44-60). Excretion of radioactivity was both by renal (32-56%) and fecal (47-52%) routes.
In vitro, terconazole is highly protein bound (94.9%) and the degree of binding is independent of the drug concentration.
Photosensitivity reactions were observed in some normal volunteers following repeated dermal application of terconazole 2.0% and 0.8% creams under conditions of filtered artificial ultraviolet light. Photosensitivity reactions have not been observed in U.S. and foreign clinical trials in patients who were treated with terconazole 0.8% vaginal cream.
Microbiology: Terconazole exhibits fungicidal activity in vitro against Candida albicans. Antifungal activity also has been demonstrated against other fungi. The MIC values for terconazole against most species of lactic acid bacteria typically found in the human vagina were >/=128 mcg/mL. The exact pharmacologic mode of action of terconazole is uncertain; however, it may exert its antifungal activity by the disruption of normal fungal cell membrane permeability. No resistance to terconazole has developed during successive passages of C. albicans.
TERAZOL 3 Vaginal Cream is indicated for the local treatment of vulvovaginal candidiasis (moniliasis). As TERAZOL 3 Vaginal Cream is effective only for vulvovaginitis caused by the genus Candida, the diagnosis should be confirmed by KOH smears and/or cultures.
Patients known to be hypersensitive to terconazole or to any of the components of the cream.
None.
General: Discontinue use and do not retreat with terconazole if sensitization, irritation, fever, chills or flu-like symptoms are reported during use.
Laboratory Tests: If there is lack of response to TERAZOL 3 Vaginal Cream, appropriate microbiologic studies (standard KOH smear and/or cultures) should be repeated to confirm the diagnosis and rule out other pathogens.
Drug Interactions: The levels of estradiol (E2) and progesterone did not differ significantly when 0.8% terconazole vaginal cream was administered to healthy female volunteers established on a low dose oral contraceptive.
Carcinogenesis: Studies to determine the carcinogenic potential of terconazole have not been performed.
Mutagenicity: Terconazole was not mutagenic when tested in vitro for induction of microbial point mutations (Ames test) or for inducing cellular transformation, or in vivo for chromosome breaks (micronucleus test) or dominant lethal mutations in mouse germ cells.
Impairment of Fertility: No impairment of fertility occurred when female rats were administered terconazole orally up to 40 mg/kg/day for a three month period.
PREGNANCY: Teratogenic Effects.
Pregnancy Category C.
There was no evidence of teratogenicity when terconazole was administered orally up to 40 mg/kg/day or subcutaneously up to 20 mg/kg/day in rats. Dosages at or below 10 mg/kg/day produced no embryotoxicity; however, there was a delay in fetal ossification at 10 mg/kg/day in rats. There was some evidence of embryotoxicity in rabbits and rats at 20-40 mg/kg. In rats, this was reflected as a decrease in litter size and number of viable young and reduced fetal weight. There was also delay in ossification and an increased incidence of skeletal variants. The no-effect oral dose of 10/mg/kg/day resulted in a mean peak plasma level of terconazole in pregnant rats of 0.176 mcg/mL which exceeds by 30 times the mean peak plasma level (0.006 mcg/mL) seen in normal subjects after intravaginal administration of terconazole 0.8% vaginal cream. This safety assessment does not account for possible exposure of the fetus through direct transfer of terconazole from the irritated vagina by diffusion across amniotic membranes. Since terconazole is absorbed from the human vagina, it should not be used in the first trimester of pregnancy unless the physician considers it essential to the welfare of the patient.
Nursing Mothers: It is not known whether this drug is excreted in human milk. Animal studies have shown that rat offspring exposed via the milk of treated (40 mg/kg/orally) dams showed decreased survival during the first few post-partum days, but overall pup weight and weight gain were comparable to or greater than controls throughout lactation. Because many drugs are excreted in human milk, and because of the potential for adverse reaction in nursing infants from terconazole, a decision should be made whether to discontinue nursing or to discontinue the drug, taking into account the importance of the drug to the mother.
Pediatric Use: Safety and efficacy in children have not been established.
During controlled clinical studies conducted in the United States, patients with vulvovaginal candidiasis were treated with terconazole 0.8% vaginal cream for three days. Based on comparative analyses with placebo and a standard agent, the adverse experiences considered most likely related to terconazole 0.8% vaginal cream were headache (21% vs. 16% with placebo) and dysmenorrhea (6% vs. 2% with placebo). Genital complaints in general, and burning and itching in particular, occurred less frequently in the terconazole 0.8% vaginal cream 3 day regimen (5% vs. 6%-9% with placebo). Other adverse experiences reported with terconazole 0.8% vaginal cream were abdominal pain (3.4% vs. 1% with placebo) and fever (1% vs. 0.3% with placebo). The therapy related dropout rate was 2.0% for the terconazole 0.8% vaginal cream. The adverse drug experience most frequently causing discontinuation of therapy was vulvovaginal itching, 0.7% with the terconazole 0.8% vaginal cream group and 0.3% with the placebo group.
Overdose of terconazole in humans has not been reported to date. In the rat, the oral LD 50 values were found to be 1741 and 849 mg/kg for the male and female, respectively. The oral LD 50 values for the male and female dog were [cong ]1280 and >/=640 mg/kg, respectively.
One full applicator (5 g) of TERAZOL 3 Vaginal Cream (40 mg terconazole) should be administered intravaginally once daily at bedtime for three consecutive days. Before prescribing another course of therapy, the diagnosis should be reconfirmed by smears and/or cultures and other pathogens commonly associated with vulvovaginitis ruled out. The therapeutic effect of TERAZOL 3 Vaginal Cream is not affected by menstruation.
TERAZOL 3 (terconazole) Vaginal Cream 0.8% is available in 20 g (NDC 0062-5356-01) tubes with an ORTHO® Measured-Dose Applicator. Store at controlled room temperature 15-30°C (59-86°F).
Caution: Federal (U.S.A.) law prohibits dispensing without prescription.
631-11-314-4 Revised March 1995
 |